Rounding out our Feature Round Up series, this article will showcase some of the new clinical capabilities of our eTMF. To read more about the overall features of Kivo GO, our new Quality Management System, or our Regulatory updates, check out the rest of the Feature Round Up. To learn about the latest updates for Clinical, continue reading!
Active Trial Management
Arguably the biggest addition to our Clinical capabilities is Active Trial Management.
Managing an active clinical trial requires a wide variety of tools, vendors, systems, and resources, from CTMS, EDC, enrollment, scheduling and more. From the sponsor's perspective, keeping track of the many moving parts can be challenging, especially for smaller sponsors with limited resources. To help these teams, we’ve added the Active Trial Management module.
With Trial Management, you can build and templatize project plans for any phase of your trial, including site selection, study startup, and closeout, as well as handle events as they occur, such as adverse events, protocol changes, supply chain issues, and more.
Trial Milestones
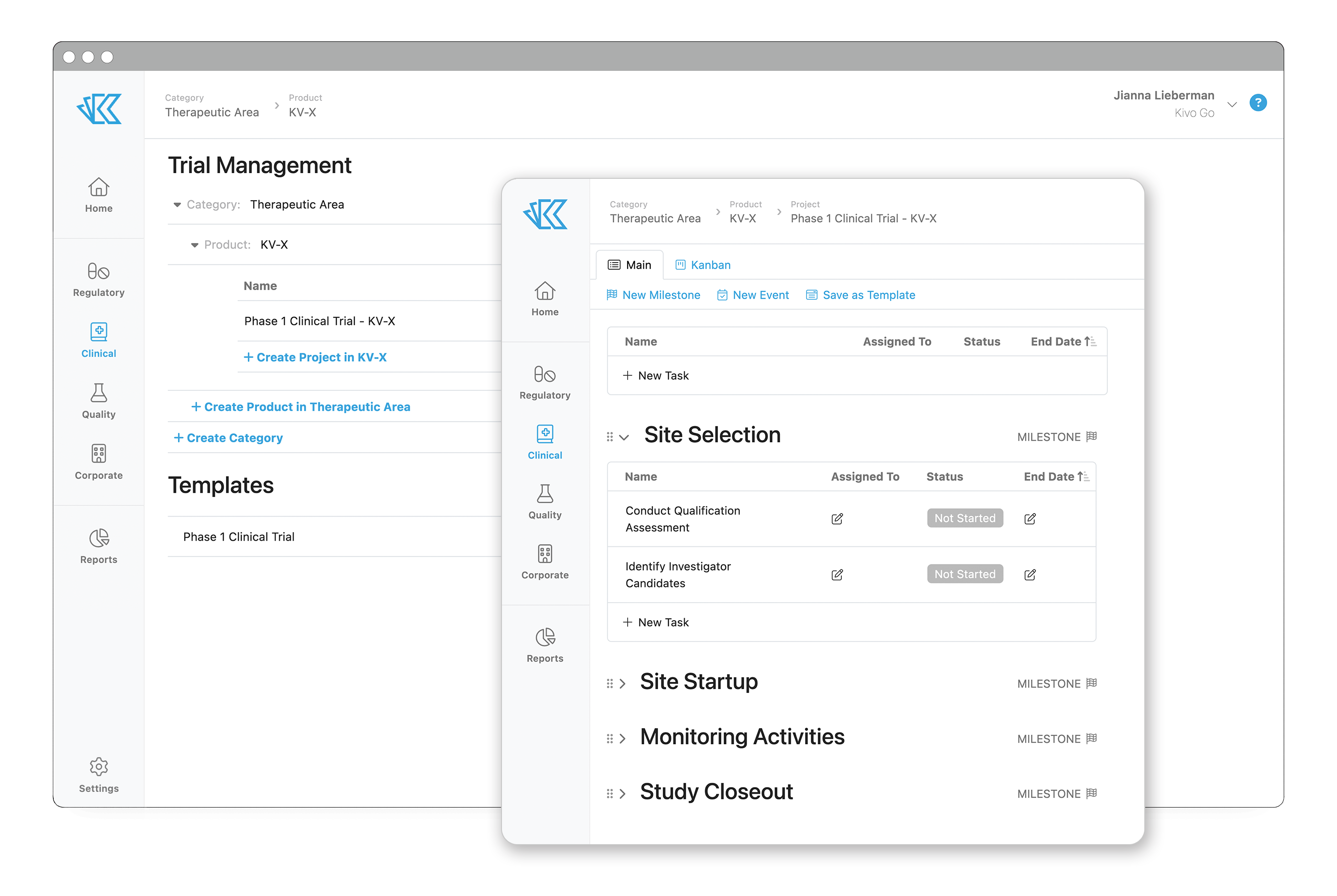
Activities like site selection and study startup typically follow a repeatable process. But the management of that process can be painful. From multiple file-sharing tools, tracking spreadsheets, manual notes and reminders, and other workarounds, there are many moments where things can fall through the cracks or slow down your overall timeline.
When you create your first study in Kivo, you’ll have the opportunity to build out a project template for these activities - or use our out-of-the-box template and customize later. Our standard template for an active trial includes Site Selection, Study Startup with a Greenlight Package, Monitoring Activities, and Study Closeout. Each of these project milestones contains the relevant tasks needed to complete each part of the project. As you add information to these tasks, such as assigned individual, start and end dates, related documents, and more, you’ll be creating your project timeline!
Adding Events
Of course, many events can (and do) occur during a clinical trial. To keep track of these events, you can add a New Event to your project timeline. Default events include Protocol, Investigator, or Monitoring Changes, Adverse Events, Supply Chain Issues, and Audits, but you can also add as many custom Events as needed for your trial. Adding an event to your trial timeline will automatically add the relevant tasks and subtasks as determined by your SOP.
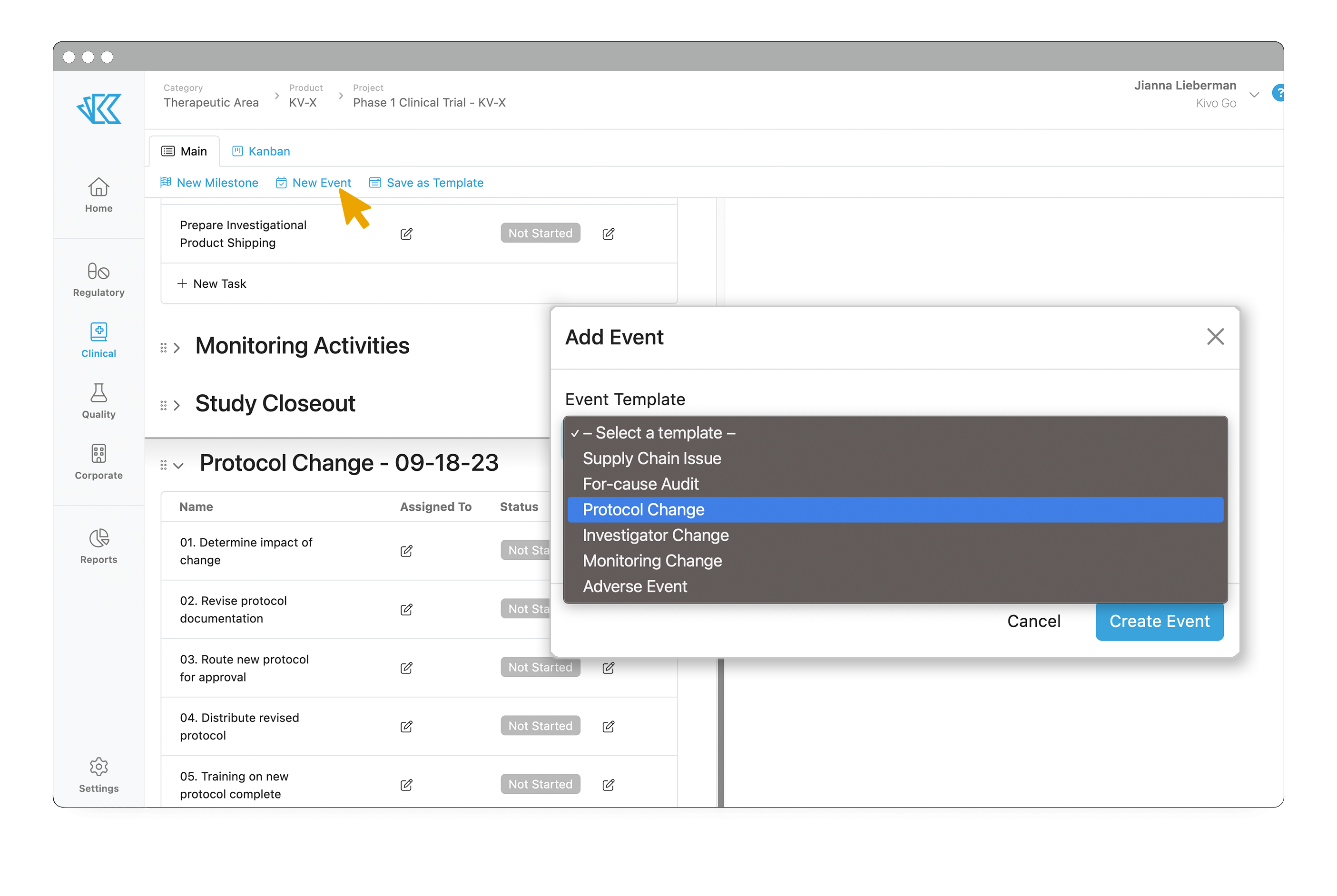
By templatizing these milestones and events, you can get up and running much more quickly. And by centralizing all the required work into one place, you have the visibility required to keep things moving smoothly. All vendors can be given access to Kivo, so they can upload documents directly into the eTMF. Every document that lives in Kivo will be logged within the audit trail, so you are preparing for inspection from the very beginning. And finally, you can run reports for any project or across projects, to easily identify bottlenecks and potential issues.
Investigator Site Management
In addition to managing your trials inside Kivo, you can also manage investigator sites. By storing information about all sites within the system, you can speed up future site selection significantly, as well as more easily manage existing sites.
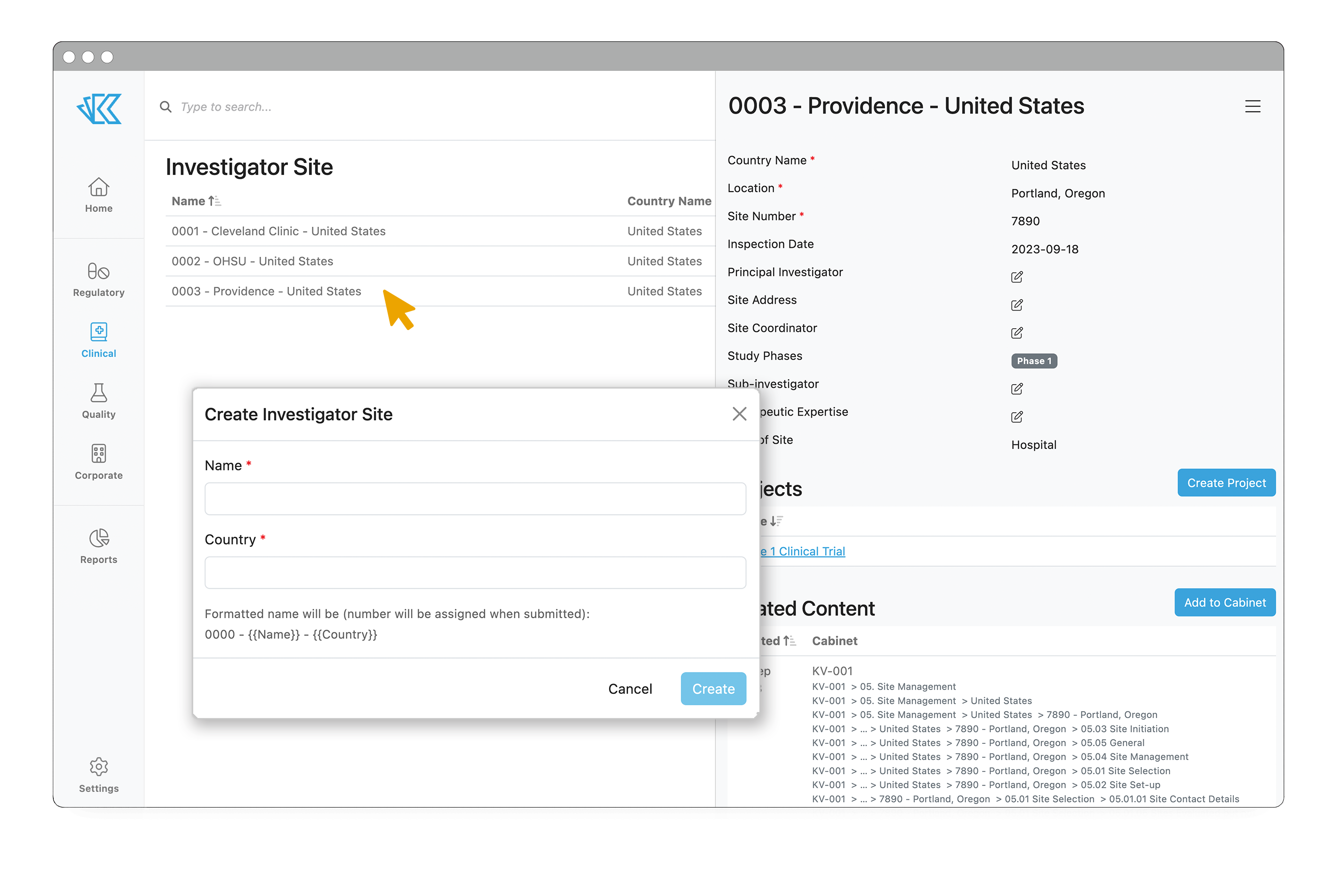
To add a new Site into the system, simply select “Create Investigator Site” and follow the prompts. The fields you can fill out are fully customizable to match your needs. Default fields include location, site number, inspection date(s), address, type of site, study phases, and more. Should you need to add an existing site to a new trial, that’s as easy as a few button clicks.
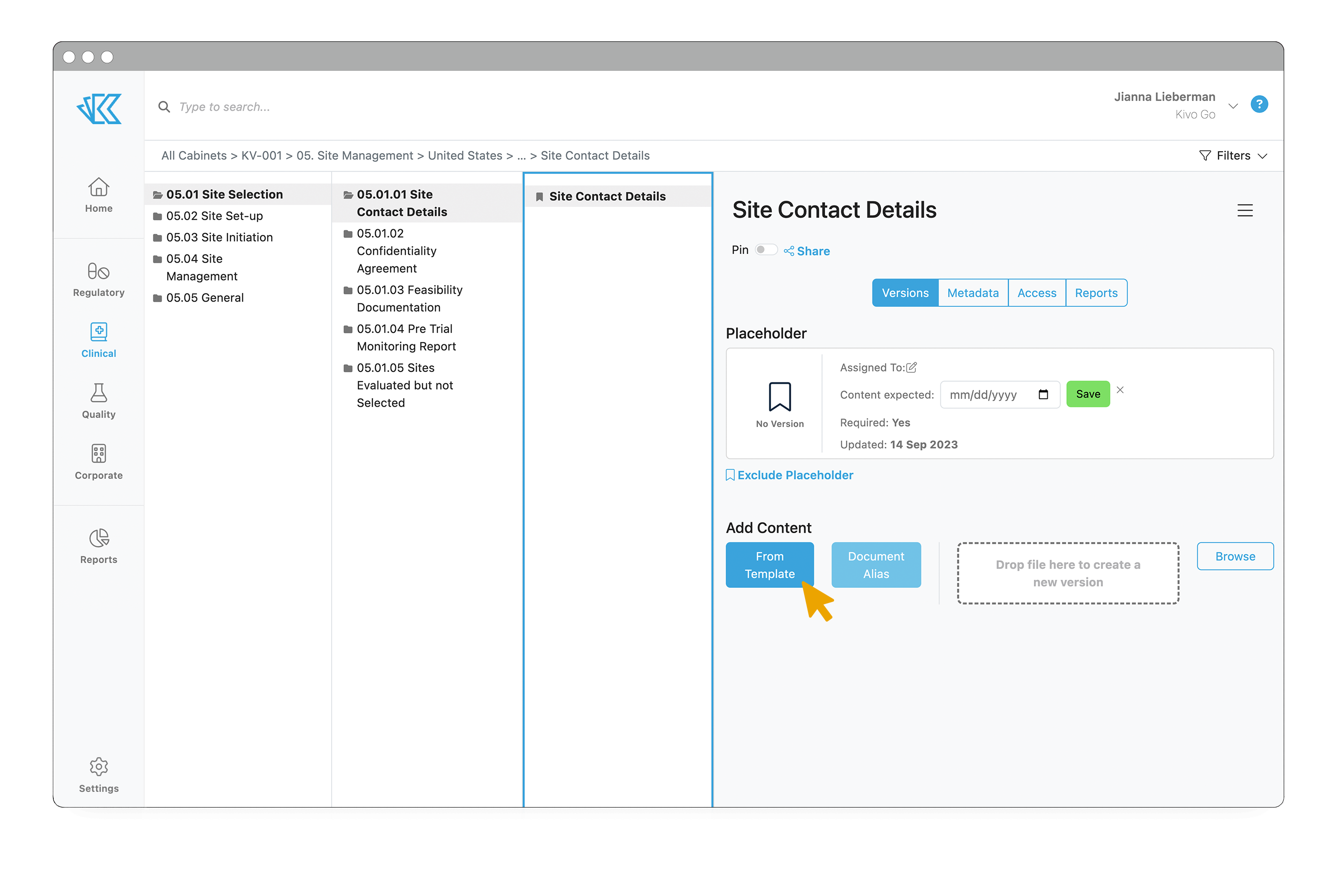
Crucially, once you have added a site, you can associate the site with any project (ie study), and add the relevant content placeholders immediately into the eTMF, with the correct file structure. This saves significant time figuring out what documentation is necessary or adding those documents one by one to a file sharing system. If you grant your vendors access to Kivo, they will be able to upload documents directly to those placeholders. You can even set up trainings within Kivo to ensure your sites have read and understood all the necessary protocols and SOPs.
Easy TMF Migration
Finally, let’s talk about transferring an existing TMF from a CRO or other platform.
There are several common reasons you may find yourself needing to transfer your TMF from one system to another. You may have scaled to the point that you’d like to manage your TMF in house rather than use your CRO’s system, you’ve reached the end-of-study and your contract requires that you take possession of your data, or perhaps you’d like to change your eTMF software vendor.
Regardless of the reason, migrating a TMF is an extremely important process that can sometimes be overlooked. If you have an upcoming TMF migration, leverage these best practices to ensure you protect your data’s integrity from start to finish. Otherwise, you may create gaps in the audit trail that throw your entire trial into question during an inspection. Should you want support for this process, inquire about our TMF Transfer Services.
To facilitate these transfers, we have created a first-of-its-kind TMF Migration Tool.
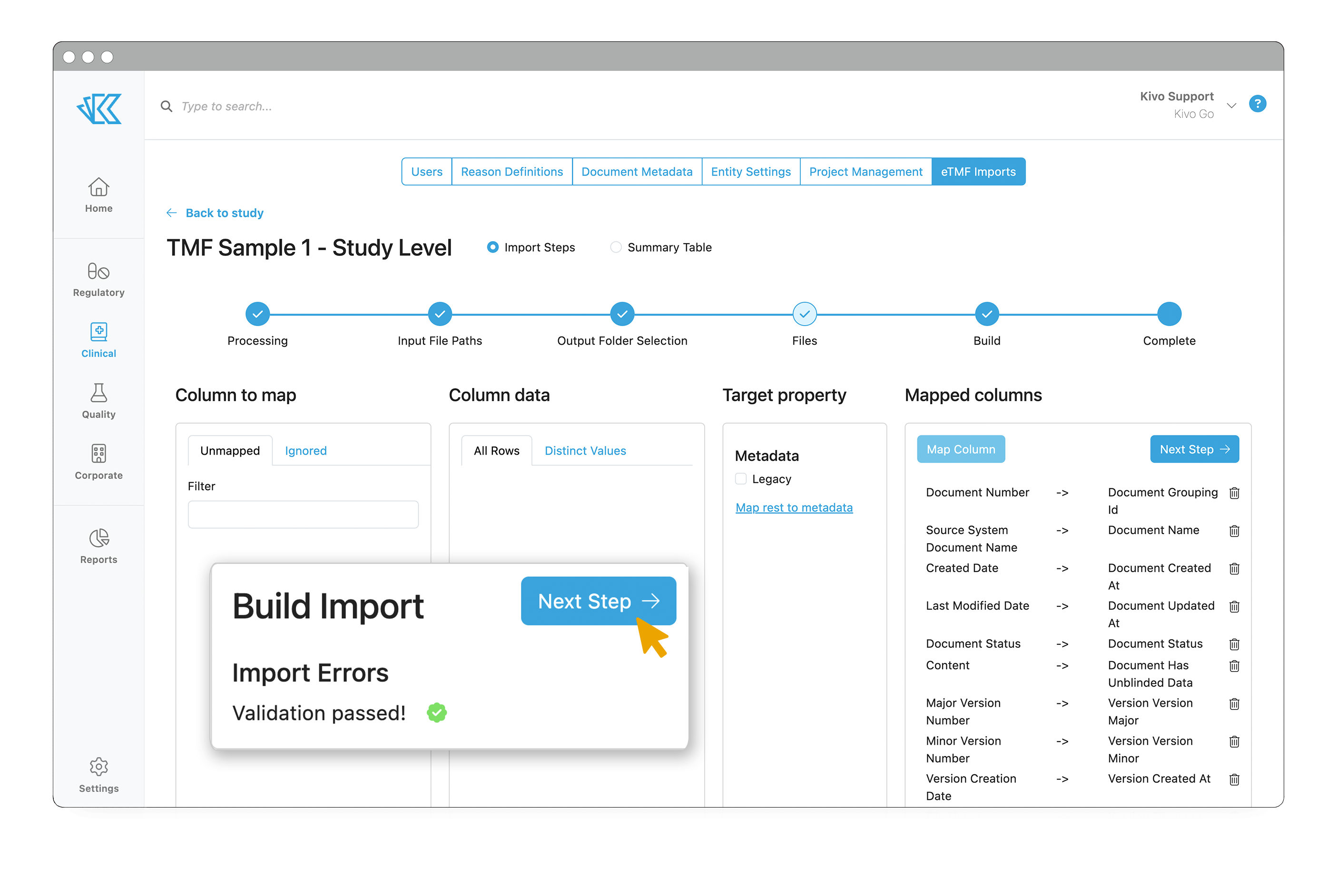
Many software and service providers handle TMF migrations manually, with labor-intensive (and error-prone) approaches that take weeks - or even months - to complete. At Kivo, we knew that we could improve this process by building an automatic (and validated) migration tool, coupled with hands-on quality control.
To initiate a transfer, we grant your CRO write-only access to our secure FTP site. As soon as they upload a file, every interaction with the data is captured in our audit trail. As we move through the import tool step by step, you have the ability to map all existing metadata into the corresponding metadata field in Kivo. If there isn’t a matching field in Kivo, we map that data to a Legacy field, ensuring no data is lost. Unique to Kivo, our TMF Migration tool also parses audit trail files from the source system and recompiles them into our audit log, so you have a single, unbroken record of each and every document - segmented by the system in which the action occurred.
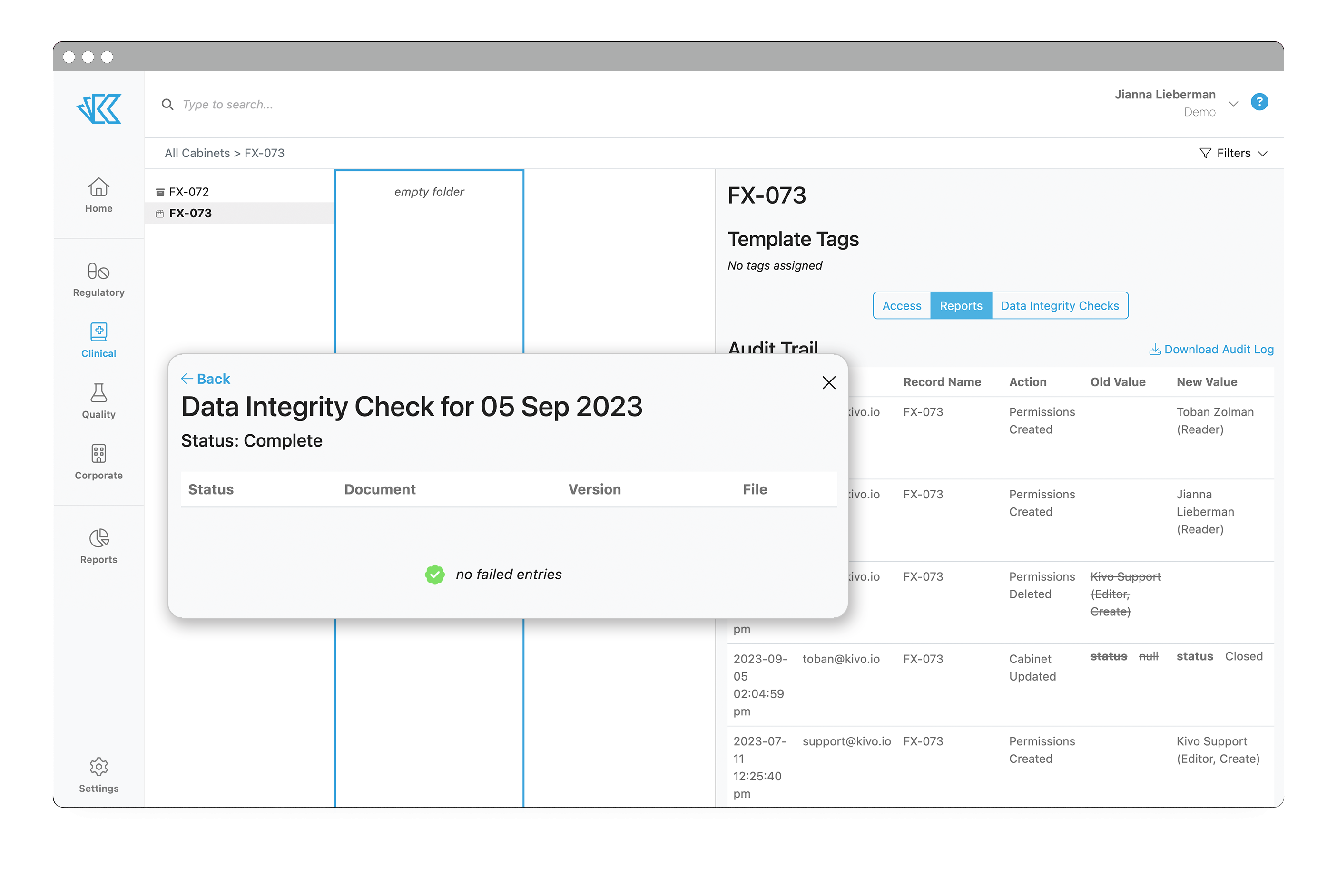
After your TMF is migrated, we conduct a manual quality check to verify all data has been imported and mapped correctly. Should you store your TMF in Kivo long term, we also run automated quarterly data integrity checks to eliminate the risk of data corruption over time.
For further information about TMF Migration, please see the following resources:
- Best Practices for End of Study Transfer (Blog Article)
- TMF Transfer Checklist (Whitepaper Download)
- 7 Questions to Ask your CRO about your TMF
- Why Not to Keep your TMF with your CRO
- Considerations for Moving an Electronic Clinical Trial
- IT Best Practices for Receiving eTMFs from CROs (Webinar Recording)
- Managing the End-of-Study TMF Transfer (Webinar Recording)
In Summary
As clinical trials become more decentralized, having a single source of truth where projects, vendors, and data can be managed becomes increasingly important. We developed Kivo because we saw an opportunity to streamline many of the manual operations and workflows of our Clinical, Regulatory, and Quality colleagues, while simultaneously improving compliance and inspection outcomes.
We united these three functions in our platform, Kivo GO. We added a QMS to support Quality operations. We beefed up our Regulatory capabilities with robust project management and the ability to track correspondence and commitments. And with these Clinical improvements, we’ve made it easier to manage active trials, investigator sites, and your TMF data. We’re deeply proud of the work we have done, and hope that it accelerates the important work sponsors are doing around the world to improve patient outcomes and care.
Finally, we believe in a culture of continuous improvement, so these will not be the last updates to our platform! We are already cooking on the next batch of new features, with the help of feedback from our clients and partners. So please keep in touch, and let us know if there is any way we can support you in your efforts.



