Compliance, Meet Simplicity.
Introducing: Kivo GO
Finally, an intuitive document management platform for scaling life science teams.
DMS | RIM | eTMF | QMS

Validated On Day 1
CFR Part 11 Compliance
Single Sign On
The world of drug development has changed
Once dominated by “Big Pharma”, today over 70% of active drug trials are sponsored by small, fast-moving teams — and speed-to-market is more important than ever.
But so is compliance. And safety. And security. Emerging life sciences teams shouldn't have to choose between efficiency and compliance. Kivo GO offers a better way to work together.

Kivo GO
One Platform for All Teams
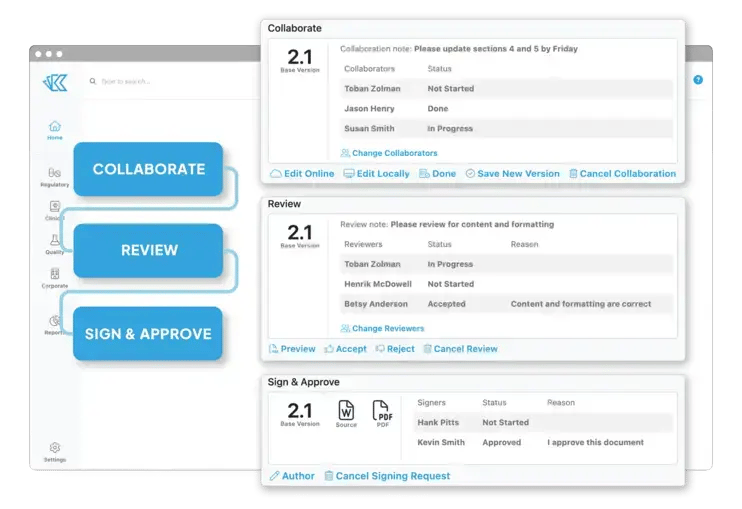
Document Collaboration
Kivo GO brings all your team's documents, data and to-do's into a unified and easy-to-use system that includes:
- An intuitive interface
- Customizable workflows
- Built-in reporting and project management
Stop chasing files and battling permissions - Kivo makes it easy to work together, efficiently.

Modules for Every Function
Drug development is a team sport between Regulatory, Clinical and Quality. Kivo has built-in tools for every team:
- RIM for submissions, INDs and publishing integration
- eTMF and CTMS features for active & archived trials
- QMS to manage SOPs, CAPAs, vendors & more
...all in one, unified system.

Compliant & Secure
In regulated industries, work works differently. Kivo was built by and for life sciences professional, and features:
- Continuous validation
- CFR 21 Part 11 compliance
- Secure, SOC 2 verified
With Kivo, you can rest assured that GCP is maintained across your teams - and with a system that's secure and scalable.
Getting Started with Kivo GO

- Kivo GO includes access to RIM, eTMF, and QMS, all for the same price.
- Documents are linked between modules, so everyone is looking at the same information. No aliases required.
- With role-based pricing and permissions, you can grant access to vendors, contractors, and investigators as needed
- Intuitive navigation makes onboarding and user training simple
- Implementation takes less than a week


- Instantly build out study and submission structures with workflow templates
- Smart document placeholders tell you where information needs to go
- Pull in your milestones and project timelines to keep everything on track
- Automatic audit trails capture every move and change in the system, so you are always audit ready
- Import existing studies and past submissions
- Tie correspondence to the appropriate submission and document


- Reduce manual work with intelligent reporting
- Automate important alerts, such as past due documents, documents that haven't been viewed by the necessary parties, or expiring vendor certifications.
- Eliminate manual tracking spreadsheets for tasks like publishing handoffs, vendor tracking, and agency commitments.
- Track project timelines via the dashboard, so you have instant visibility into your status

Everything is included, for everyone who needs it
Kivo GO is designed (and priced) for the way growing sponsors do business.
Annual Subscription
Role-based per user pricing for Full Platform, Limited Access, and 3rd-Party Access
No Hidden Fees (Seriously)
Say goodbye to unexpected fees with Kivo, including:
Trusted by Sponsors Worldwide
With decades of experience in both regulatory and cloud-based software,
Kivo offers best-in-class support and security, tailored specifically to the life sciences industry.
Doubling Programs in 2 Years: How Hyloris Scaled Clinical, Regulatory and Quality with Kivo

Real Human Support
- Sub-5-Minute Response Time
- Unlimited Support Included
- Unlimited User Training
Best-in-Class Security
- SOC 2 Compliant
- Fully Validated
- CFR Part 11 Compliant
- Quarterly data integrity checks
- Single Sign-On - No IT Required
Implement Within Weeks
- No Implementation Fee
- No Validation Fee
- Up and Running Within Days
What our customers are saying
Recent Reviews

Kristi Norris
VP of Regulatory Affairs
"Kivo was really fast to set up training, and then because the system is so user-friendly, it just didn’t take that much time to get people up to speed. Normally the learning curve with a system like this is quite steep, but Kivo is so intuitive we can quickly get people oriented and off to the races."

Seppe G.
Director of Regulatory Affairs and Quality Assurance
"The intuitive on-line interface has been adapted quickly by the entire development team allowing both the CMC experts as well as the clinical team to seamlessly integrate internal documents as well as controlled documents from external parties."

Veronique V.
Quality Assurance Manager
"We're very happy to have all our documents stored in the same place. It's efficient to have a system where the review of a doc can be done simultaneously by several reviewers. The Kivo team is always ready to help and always listen to customer needs. Really appreciated."
Accelerate Your
Workflow with Kivo
See how Kivo can help your team reduce manual work and accelerate speed-to-market.



