REGULATORY PROJECT MANAGEMENT
Easily Manage Regulatory Projects & Submissions
Give your entire team easy, controlled access to your submission or dossier documents - and organize their work together with Kivo's Project & Dossier Management System
Project Management Tools Tailored for Regulatory Professionals
Kivo makes it easy to manage your submissions with control and visibility across every document, folder, binder and dossier.
Managing submissions and regulatory projects is complicated by nature, but it can be streamlined via a thoughtful combination of project management, reporting and intelligent automation. Kivo was built by and for regulatory experts to simplify the management of these complex submission processes and maintain compliance at every step.
PROJECT AND DOSSIER MANAGEMENT
Less time chasing documents means less deadline stress
Because every document related to your submission project or dossier resides in Kivo’s intuitive Document Management System - everyone on your team has access to everything they need, when they need it. Not only are those same documents then available to Kivo’s eCTD Viewer and Submission Builder - but also to our project management and reporting tools - allowing you to easily track and manage submissions.
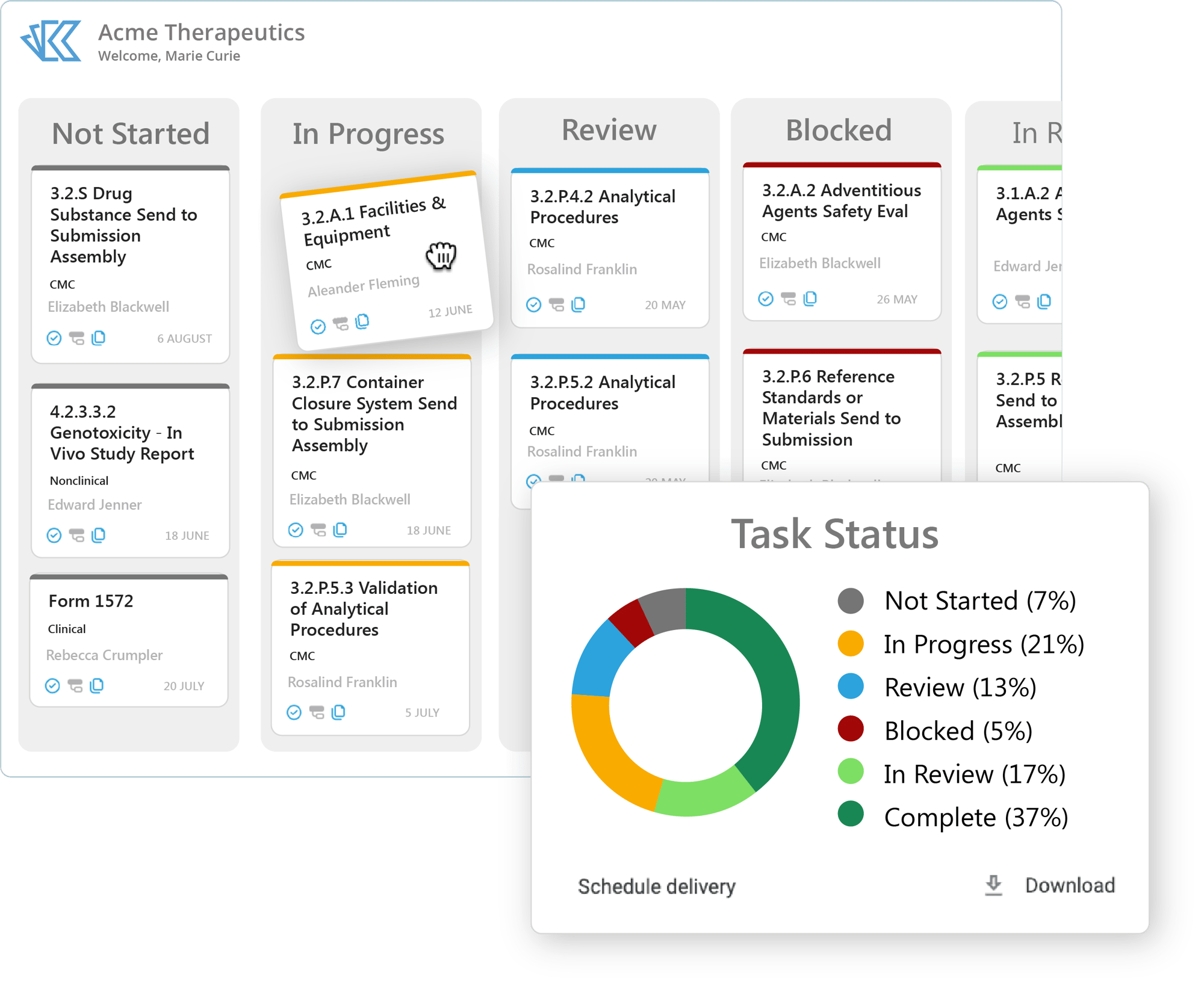
Kivo's Regulatory Project Management Features Include:
Kanban View
Easily drag and drop documents from one customizable status to the next. Status is color coded for viewing at a glance.
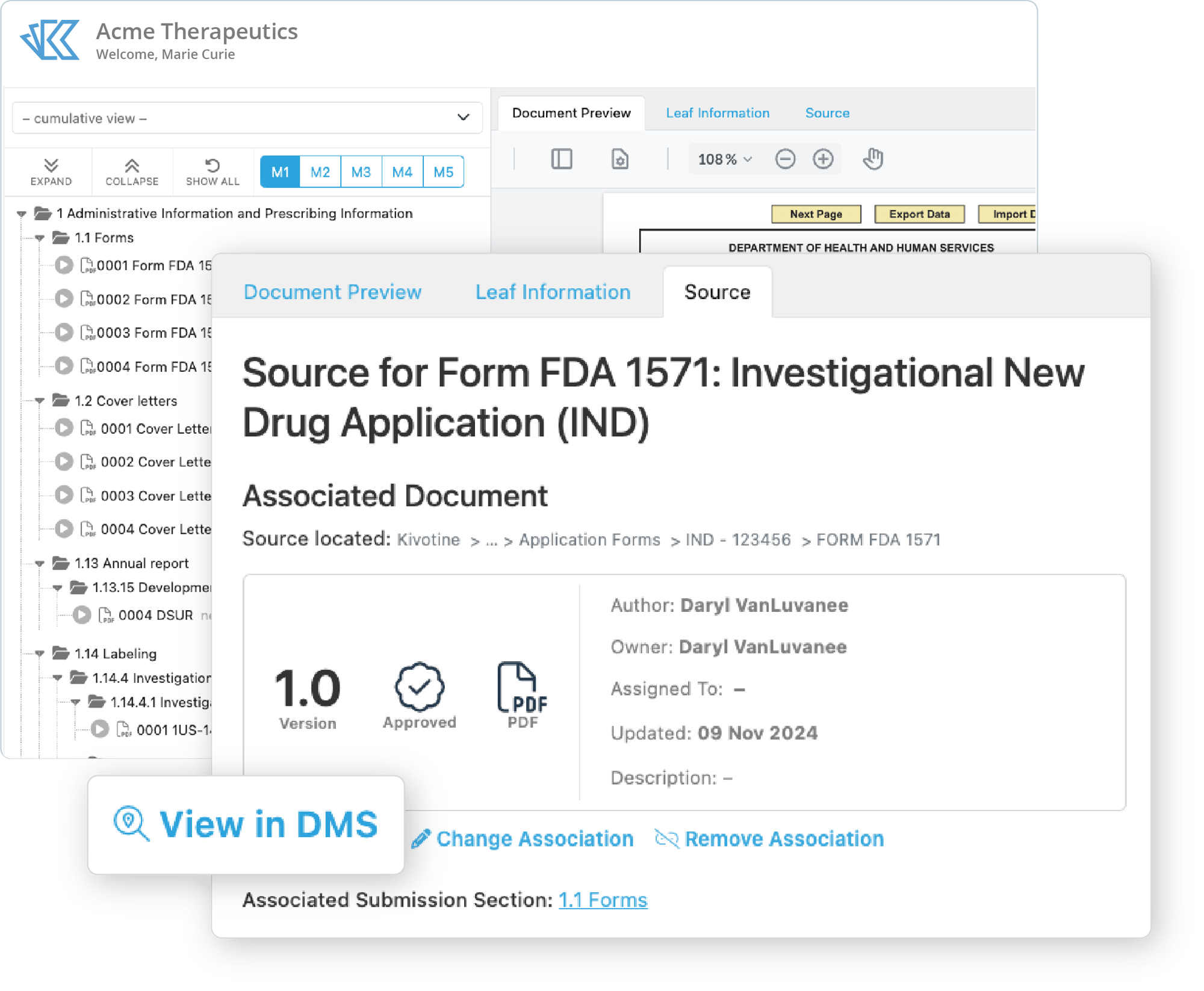
Link Content to the DMS
All documents reside directly in our DMS so you have instant access to every version, all metadata and audit trails
Project Tasks & Milestones
Build task templates for common project types that align with your SOPs. Add dependencies to gain visibility into timelines and potential blockers.
Correspondence & Commitments
Tie correspondence back to the project with metadata. Add commitments as needed, and build reporting on commitments by agency or timeframe.
Intelligent Reporting
Run reporting by user, vendor, document, project, submission...the list is (almost) endless. Then automate the reports you need to deliver straight to your inbox.
Controlled Access
Easily grant or revoke access to submissions at any time, from internal team members to third party vendors to your publisher, speeding up collaboration while maintaining compliance.
Accelerate Your
Workflow with Kivo
See how Kivo can help your team reduce manual work and improve compliance.


Kivo FAQs
Absolutely. While spreadsheets are familiar and flexible, they fall short when it comes to version control, team collaboration, and traceability. They’re hard to maintain, easy to break, and don’t scale well as your submissions grow in complexity.
Kivo replaces disconnected spreadsheets with a real system purpose-built for regulatory work. You still get the flexibility to customize timelines, structures, and workflows, but everything is tied to your actual submission content and tracked in a compliant, auditable way. It’s faster, safer, and much easier to manage.
A dossier management system is a specialized software solution used by life sciences companies to collect, organize, manage, and track all documentation required for regulatory submissions. Whether you're submitting an IND, NDA, MAA, or 510(k), a dossier includes hundreds (sometimes thousands) of documents that must meet strict formatting, versioning, and traceability requirements. A dossier management system ensures that every document is controlled, approved, and submission-ready, with proper metadata, access controls, and audit trails in place.
For regulatory teams, this is mission-critical. Without a structured system, teams often rely on email threads, shared folders, and spreadsheets, which can quickly lead to version confusion, missed updates, and compliance risks. A dossier management system like the one offered through Kivo centralizes the submission content, supports collaborative workflows, tracks document readiness, and ensures that what gets submitted is exactly what was approved. In short, it transforms regulatory chaos into a controlled, auditable process.
Coordinating submissions across multiple markets is a logistical challenge. Each agency has different requirements, formats, and deadlines. A good regulatory project management system should let you track and manage those timelines without duplicating work or losing sight of dependencies.
With Kivo, you can create parallel or staggered submission timelines for each region while reusing shared content and tracking market-specific requirements. Teams can see which modules are complete, which are pending updates, and what needs to change to meet local requirements. You get global visibility without siloed trackers or duplicated documents.
You should be able to, but not every system is built with regulatory needs in mind. Generic project tools let you assign tasks, but they often don’t include regulatory workflows like controlled review, approval, and electronic signatures.
Kivo lets you assign owners to specific documents, define due dates, and route each file through a compliant review and approval process. Approvals are tracked with electronic signatures, full audit trails, and Part 11 compliance. You’re not just checking off tasks, you’re creating a documented, defensible process.
Most of the tools advertised for regulatory project management are simple task trackers. The best software goes deeper, designed for the complexity of global submissions, traceability requirements, and the need to align cross-functional teams. Life sciences companies need more than Gantt charts or Jira-style boards. They need a system that ties regulatory plans directly to the content, approvals, and compliance workflows that drive submissions forward.
Kivo was built for exactly this purpose. Unlike generic PM tools or rigid enterprise platforms, Kivo provides structured yet flexible project tracking tied directly to your DMS and RIM system. Teams can define their submission plan, assign responsibilities, monitor content status in real time, and generate submission-ready outputs within a system that’s Part 11 compliant and purpose-built for regulatory use.
Kivo empowers regulatory and clinical project managers by bringing together content, timelines, and team accountability into one unified platform. Instead of juggling spreadsheets, shared drives, and disconnected PM tools, project managers can oversee submission progress directly within the system that houses the actual documents. With Kanban views, Gantt-style timelines, and task templates purpose-built for life sciences workflows, Kivo makes it easy to assign responsibilities, track progress, and keep cross-functional teams aligned.
Beyond tracking tasks, Kivo provides real-time visibility into the true status of each document, not just whether someone marked it “in progress.” Because Kivo integrates task management with document lifecycle workflows, project managers always know which content is still in drafting, what’s awaiting review or approval, and whether submission modules are on track. With built-in reporting, version control, and compliance features like Part 11-ready e-signatures, Kivo helps project managers deliver complex submissions on time and in full compliance, without chasing updates or risking versioning errors.
Yes, Kivo is designed to handle large-scale, complex regulatory submissions with a fully integrated approach to regulatory project management. Whether you're managing an IND, NDA, BLA, MAA, or multiple global submissions in parallel, Kivo combines submission planning, content tracking, and document management in a single, validated system. Project managers can define submission timelines, assign owners, and track readiness across all modules and markets.
With built-in templates for different submission types, role-based access controls, and real-time reporting, Kivo helps teams collaborate efficiently and stay audit-ready. You can monitor document status, manage cross-functional workflows, and generate export-ready packages without switching between disconnected tools. By embedding regulatory project management directly into the submission process, Kivo gives teams the control, visibility, and scalability needed to deliver complex submissions on time and in full compliance.
Kivo maintains compliance throughout the regulatory project lifecycle by embedding control, traceability, and auditability into every step of the process. Every action, from document creation and editing to review, approval, and export, is logged in a complete audit trail that meets 21 CFR Part 11 and GxP requirements. Version control is automatic and enforced, so teams always work from the correct document without the risk of outdated or duplicated files. Every document is linked to its metadata and approval history, giving regulatory project managers full transparency into the status and history of each submission artifact.
In addition to document-level controls, Kivo also applies compliance rigor at the project level. Task assignments, due dates, approvals, and role-based access are all tracked and enforced within the system, ensuring that every stakeholder sees and interacts only with the content relevant to their role. This helps reduce risk, improve inspection readiness, and streamline internal and external audits. To learn more about Kivo’s validation approach, security protocols, and compliance posture, you can visit our Trust Center.
Kivo’s regulatory project management tools are purpose-built for the unique needs of life sciences teams, not retrofitted from generic project management platforms. We tie project milestones, tasks, correspondence, and submission content directly to your underlying regulatory documents within a single, validated system. That means project managers aren’t just tracking progress in abstract terms; they’re monitoring real-time status tied to the actual work being done, with full visibility into which documents are approved, pending, or at risk.
Unlike most tools that require separate systems for project planning, content management, and compliance tracking, Kivo brings it all together in one seamless workflow. You can manage submission timelines, assign responsibilities, track agency communications, and generate up-to-date reports without ever leaving the platform. Because Kivo was designed specifically for regulatory professionals in pharma, biotech, and med device companies, it supports your existing processes while providing the structure and traceability required for GxP compliance and audit readiness. It’s more like a regulatory command center than a simple project management tool.

