KIVO RIM SYSTEM
The #1 Regulatory Management System for Life Sciences
The only RIM software with real-time collaboration, lifetime validation & no hidden fees.
Team plans start at $1800/month.

Simple, Compliant, Affordable RIM Software
Improve compliance & streamline regulatory operations without breaking the bank.
Regulatory operations is, at its core, process management. Your processes, and the tools you use to manage them, can either accelerate or hinder your ability to meet submission deadlines with confidence. Kivo’s RIM System is designed to help you control not only your documents but also your processes, keeping you in compliance and on track.
Validation Included
CFR Part 11 Compliant

SOC2 & ISO 9001
For every stage of the submission lifecycle
Kivo’s RIM software lets you manage all of your key regulatory functions in one easy-to-use system that everyone can access. And unlike a ‘do-it-yourself’ file-sharing solution, Kivo is Part 11 Compliant, built on the EDM Reference Model/GxP Standards, is SOC2 Certified - and even includes eSignature via DocuSign.
Support for INDs, CTAs, BLAs, NDAs, IDMPs, ANDAs, CTIS, and more.

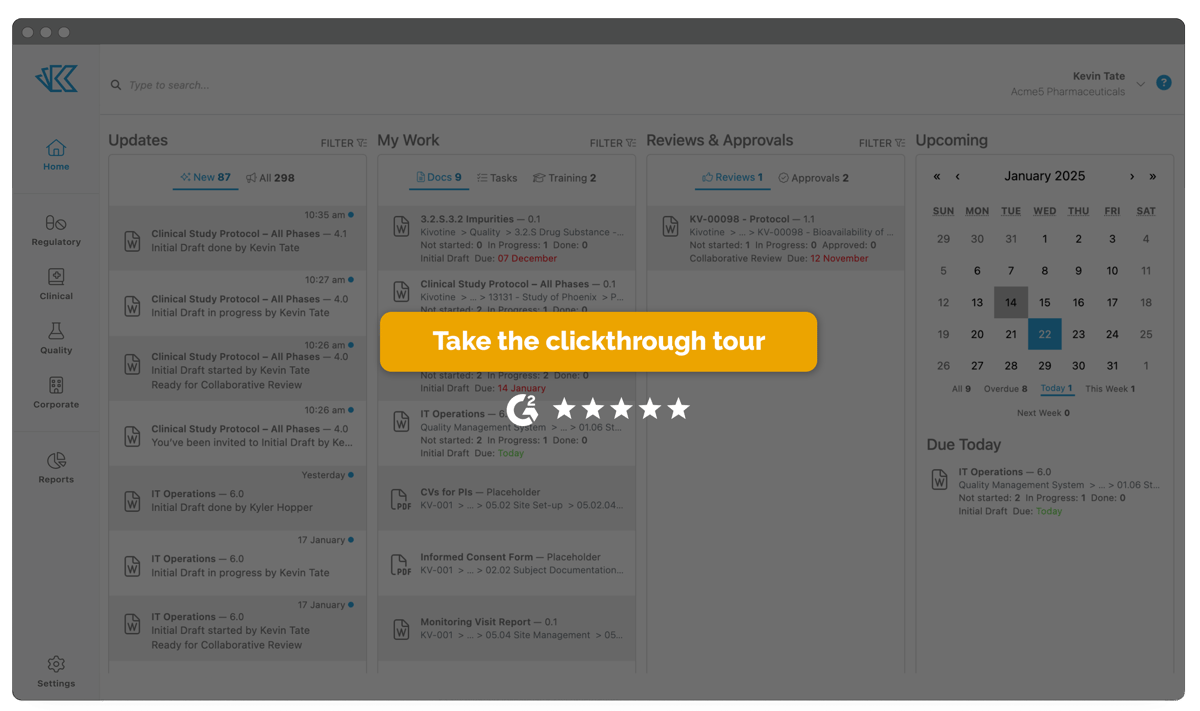
Quickly Organize All Your Documents
Manage all your controlled documents - from corporate content to submission materials to agency correspondence - inside Kivo's DMS, based on the Electronic Document Management (EDM) reference model. Easily import, view, and archive dossiers into Kivo with full traceability.
- Easy workflow for collaborative authoring, review & approval
- Part 11 Compliant eSignature with DocuSign
- Embedded PDF conversion
- Simple user permission & access management
- Over 450 pre-formatted ICH document templates
- Correspondence filing, including emails & attachments
- Automated reports to track document status, agency commitments, and project progress
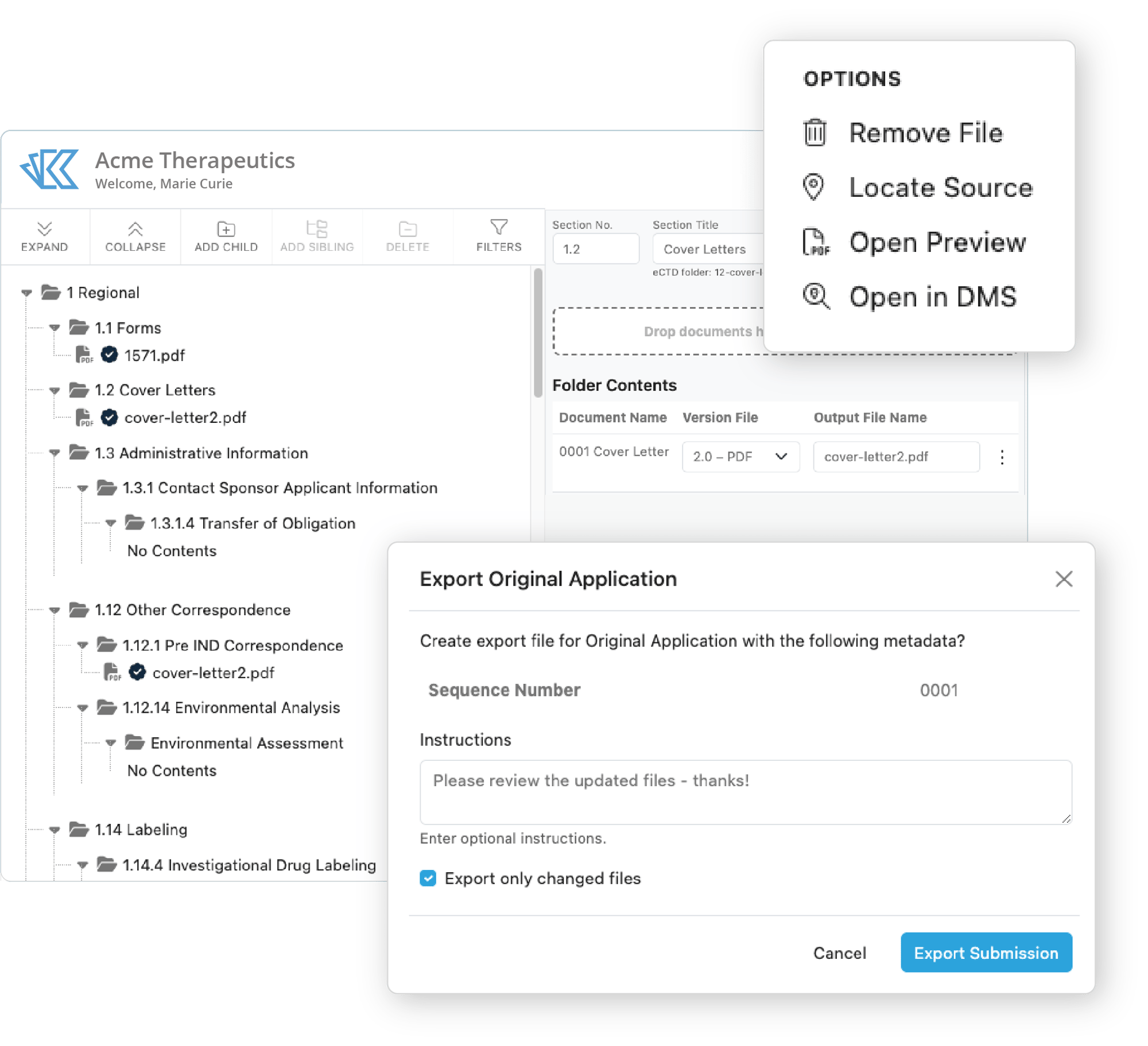
Build Submissions & Handoff for Publishing
Kivo's regulatory document submission builder and publishing preparation workflows are designed for ease of use. From your first IND to your NDA, Kivo’s RIM platform accelerates the submission lifecycle while up-leveling your compliance.
- Pre-built submission structures aligned to agency guidelines
- Documents link directly to the Kivo DMS
- Placeholders to organize around expected content - single & multiple documents
- Simple toggle for blinded and unblinded data
- Automatic tracking spreadsheets for publishing handoff
- One-click export to your publishing partner or software
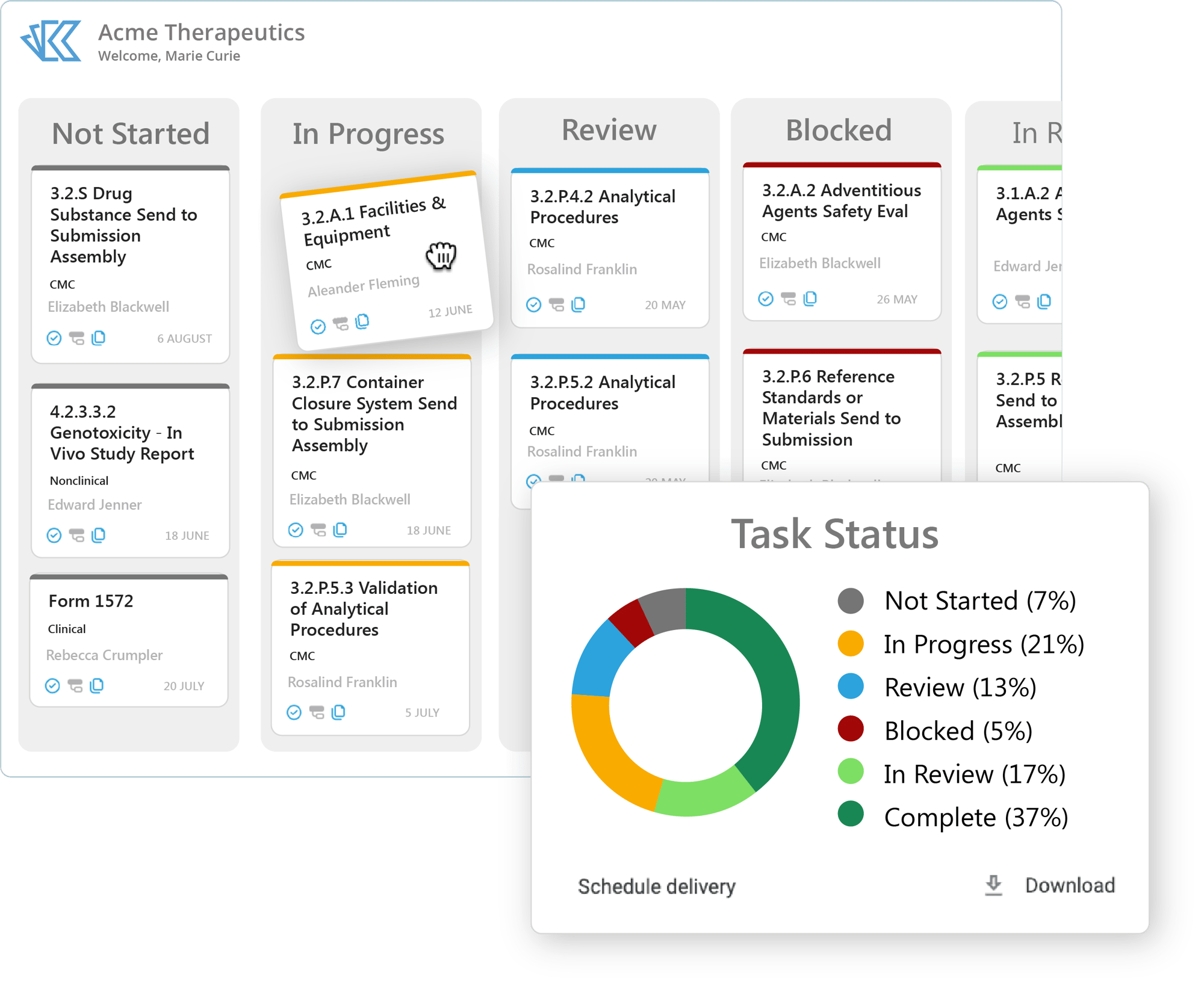
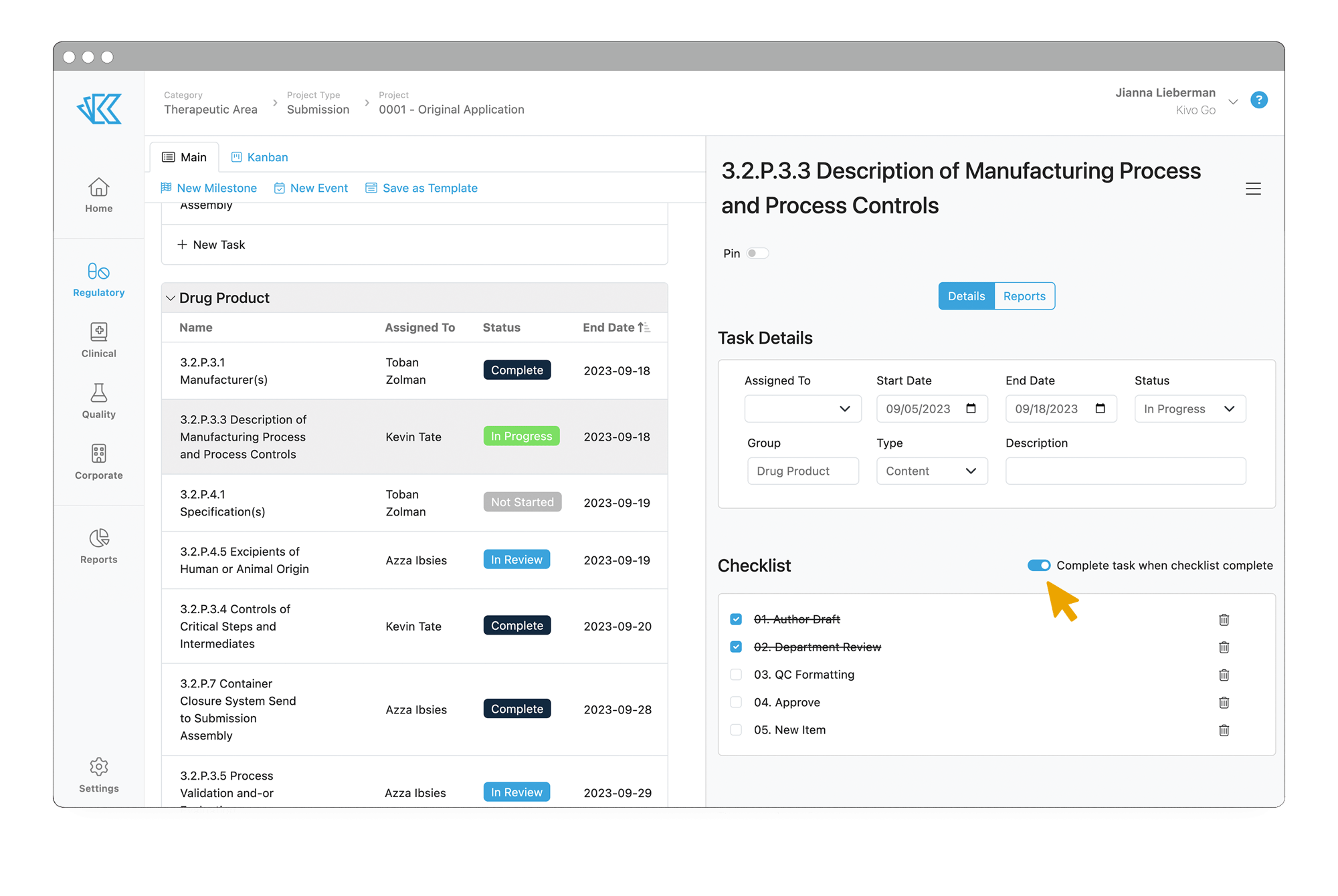
Keep Track of Your Team's Work
Never hear the phrase “manual tracking” again. With Kivo RIM, you can automate and templatize your content and submission planning, so you know at a glance if you are on track to meet your submission deadlines.
- Templatize your content plans and regulatory projects
- Assign due dates, task owners, dependencies, and sub-tasks
- Re-flow timelines automatically as needed according to due dates and dependencies
- Access real-time data directly within RIM reports
- Schedule reports to deliver automatically to your email
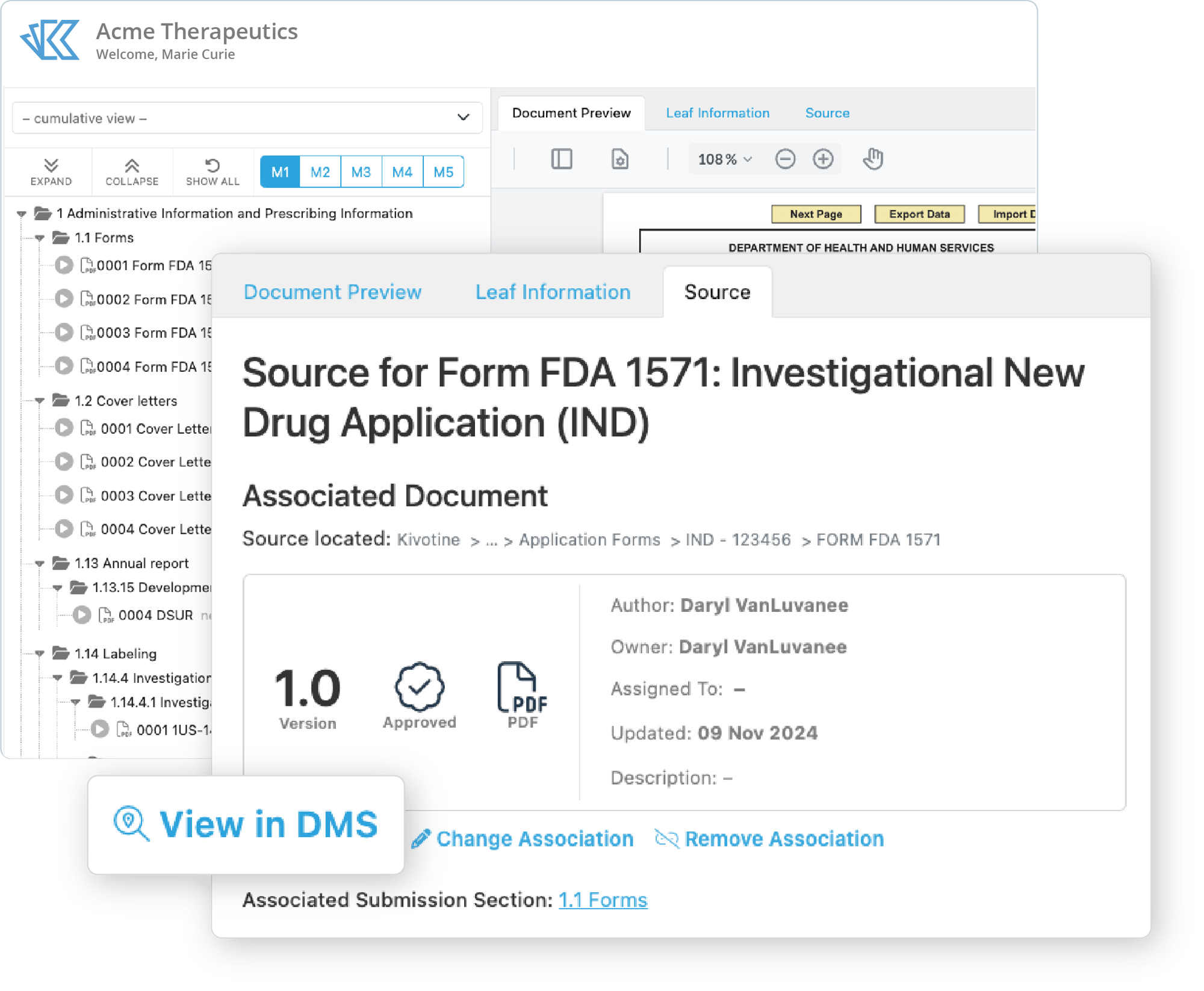
Easily View & Archive eCTDs
Locate the right version, right away. Reduce follow-up time and gain visibility into the full submission lifecycle.With Kivo's eCTD Viewer - included with any Kivo subscription - you can easily locate the precise document or correspondence that you need.
- Designed to support eCTD 4.0
- View individual sequences, current, or cumulative views
- Easy module navigation
- Inline study tagging files
- View metadata from the DMS
- Maintain submission archives within the DMS for increased control
GETTING STARTED WITH KIVO
Implementation & Migration Made Easy
Set Up Kivo in Just 2 Weeks:
- Step 1: Talk with our setup team about how you’d like your Regulatory information organized. We start with the EDM Reference Model - but our RIM software is fully customizable.
- Step 2: We’ll use our SFTP site and Data Migration tool to pull your documents, submissions, and correspondence into Kivo - structuring them to your specification, and can even preserve relevant metadata & merge audit history.
- Step 3: Kivo loads validation evidence into your workspace and can supply turn-key system management SOPs so that you can go live with compliant procedures and validation records.
- Step 4: Take a tour of your new Kivo system - make sure it all looks perfect.
- Step 5: After a 30-minute virtual training session, you’re ready to go. And Kivo’s support team is always available to help.

Best-in-Class Security & Compliance
Kivo’s team spent has spent decades working with teams of all sizes on their Regulatory submissions, projects and systems. Today, Kivo is trusted by more than 100 sponsors and service providers around the world, managing hundreds applications to the FDA, EMA, MHRA, Health Canada & more.
- SOC 2 & ISO 9001 Compliant
- FDA 21 CFR Part 11 Compliant
- Aligned to International Council for Harmonisation, EU Annex 11, ISO 27001, GxP and GCP Guidelines.

Powering Fast-Growing Pharma Teams Around the Globe
Kivo works with more than 100 sponsors & service providers in the U.S., Europe & Asia. Our solution currently manages over 100 active drug applications.
Features that scale. Costs that don't.
-
RIM FeaturesBuilt on DMS Reference ModelRole-Based, Per User Licensing for Full Platform, Limited Access, and 3rd-Party AccessSmart Document PlaceholdersDrag and Drop Document UploadCustomizable Authoring, Review, QC, and Approval WorkflowsCollaboration Features with Customizable PermissionsAutomatic Audit TrailOver 400 Pre-Formatted Templates Available (Add-On)
-
General Kivo FeaturesAccess to eTMF, eCTD and QMS Included (No Integration Required)Intelligent Analytics and Reporting with Customizable AlertsDocument Linking with Aliasing (No Multi-Indexing Required)Seamless Integration with Microsoft Office OnlineCustomizable Metadata Across Features
-
IT & SecurityIntegrated CFR Part 11 Compliant eSignaturesNo IT Required (Fully Cloud Based)Single Sign OnFully Browser Based, Available for PC and Mac
-
SupportLightning Fast Support During Business Hours (Under 5 Minutes!)Unlimited Live TrainingImplement Within WeeksUnlimited Support
What our customers are saying
Recent Reviews

Kristi N.
VP of Regulatory Affairs
"Kivo was really fast to set up training, and then because the system is so user-friendly, it just didn’t take that much time to get people up to speed. Normally the learning curve with a system like this is quite steep, but Kivo is so intuitive we can quickly get people oriented and off to the races."

Seppe G.
Director of Regulatory Affairs and Quality Assurance
"The intuitive on-line interface has been adapted quickly by the entire development team allowing both the CMC experts as well as the clinical team to seamlessly integrate internal documents as well as controlled documents from external parties."

Marissa B.
Senior Manager, Regulatory Affairs
"I have been using Kivo daily for over 2 years to manage 15+ drug development programs. It was extremely easy to learn and very intuitive for a regulatory affairs professional. Kivo makes the process incredibly easy."

Brian A.
Senior Manager, Regulatory Affairs
"Kivo is extremely user friendly, which has allowed implementation of the RIM system seamlessly throughout our company. The Kivo team has been great in walking us through the various questions we have and implementing feedback to better enhance our experience."

Lisa V.
Chief Operating Officer
"The system eliminates excel spreadsheets and other trackers, providing a great summary view into content and submission planning, preparation, and completion. It's a "must have" for small companies preparing their first IND and scales well for small and mid-sized companies looking to prepare NDAs/BLAs/501Ks."
Reduce Manual Work and Improve Compliance with the Easiest-to-Use RIM
“I can’t even express how refreshing the ease of use is, because the complexity of the traditional Part 11 Compliant systems is nonsensical. Those were designed by and for large organizations. But Kivo makes it so easy.”
-Head of IT, Elevar Therapeutics

Kivo RIM FAQs
1. What is regulatory information management (RIM) software and why is it important?
Regulatory Information Management (or "RIM") involves the systematic handling of regulatory data and documents throughout their lifecycle in industries such as pharmaceuticals, biotechnology, and medical devices. RIM software provides a central, cohesive system for life sciences teams to manage that lifecycle of regulatory activities, from document creation and submission planning to correspondence tracking and health authority commitments. It's designed to replace disconnected tools like spreadsheets, email threads, and SharePoint folders with a centralized, compliant, and collaborative system.
RIM software helps life science organizations efficiently manage regulatory obligations, reduce risks, and ensure timely product approvals while maintaining compliance with applicable laws and regulations. Without a RIM solution, regulatory teams face increased risk of missed deadlines, version control issues, and costly submission delays. With software like Kivo RIM, companies gain speed, visibility, and control, making it easier to stay compliant while accelerating time to market.
2. When should a pharma or biotech company implement a RIM system?
Most teams start evaluating RIM software as they approach or enter clinical trials. That’s when the volume of submissions, agency correspondence, and cross-functional coordination becomes too complex for spreadsheets and siloed tools.
If your team is manually tracking commitments, juggling multiple regions, or struggling to scale your submission process, it's time. The earlier you invest in RIM, the easier it is to stay compliant and audit-ready as you grow.
3. What is the best RIM software for life sciences teams?
While we are obviously biased in considering Kivo RIM to be the best RIM software for life sciences, the reality is that the most popular legacy systems in this industry are exploiting decades of branding rather than keeping up with the real needs of their users. These systems are difficult to learn, extremely complex and buggy, have slow customer support, and cost 10x more than newer, better options like Kivo that are still building a brand reputation.
Kivo RIM offers the same essential functions as these legacy systems, but unlike those systems, our software is incredibly easy to learn, designed for modern needs like combination product development, supported with 7-minute response times, and costs a fraction of the price. Our customers agree that Kivo RIM is the best RIM software for life sciences teams, and we think you will too. Click here to schedule a demo.
4. What features should I look for in a RIM system for global submissions?
When dealing with global submissions, your RIM system needs to support submission planning across multiple regions, track health authority interactions, and provide real-time visibility into the status of every activity. It should also allow you to manage country-specific requirements and keep everything aligned with agency expectations.
Critical features include eCTD viewing, correspondence tracking, dossier management, configurable workflows, and integrations with your document control and quality systems, all of which are provided readily and easily within Kivo RIM.
5. How much does RIM software cost?
Legacy RIM platforms often require six-figure budgets (with thousands in hidden costs), months of implementation, and advanced training for everyone on your team. In contrast, Kivo RIM offers team plans starting starting under $1,000/month with transparent, all-inclusive pricing, and can be implemented in less than two weeks, with everyone on your team learning the software quickly and easily, regardless of their technical background.
The cost of Kivo RIM is determined by the # of licenses you need, so be sure to visit our pricing page to learn more, but we pride ourselves on offering the best features in the industry at a shockingly affordable price. Your team shouldn't need an enterprise budget to get enterprise-grade RIM functionality.
6. Can RIM software integrate with our QMS, CTMS, or ERP?
Absolutely, and Kivo RIM makes it easy. Our platform is designed with seamless integration in mind, whether you're connecting to your QMS, CTMS, eTMF, ERP, or other critical systems. With Kivo, you can streamline data flow across your regulatory, quality, and clinical operations, eliminating manual work, reducing risk, and ensuring nothing slips through the cracks.
We offer Single Sign-On (SSO), APIs, and GxP-compliant data import-export, so your systems stay fully connected, compliant, and audit-ready from day one. No messy workarounds, no data lock-in. Just clean, secure data management that scales with you.
7. How does a RIM system help with eCTD submissions and tracking?
While many legacy RIM platforms claim to support eCTD workflows, they often rely on clunky interfaces and disconnected document tools, or they require heavy customization to create compliant structures. You may end up doing more manual work, juggling add-ons, or needing external publishing tools to get across the finish line.
Kivo RIM was built with submission-readiness at its core. Our RIM software takes the complexity out of eCTD submissions by giving your team a smarter, faster way to manage every step of the process. With pre-built structures for INDs, CTAs, NDAs, etc., you can move from draft to submission-ready with confidence.
Kivo RIM's built-in eCTD viewer lets regulatory operations teams review and validate submissions directly in the platform. You get full visibility, version control, and peace of mind, all in one intuitive system that’s built to scale with your pipeline.
8. Does Kivo RIM include Publishing?
Kivo very intentionally does NOT not include publishing. Many RIM platforms claim to offer publishing capabilities, but what they’re actually doing is white-labeling external publishing tools and bundling them into their product with a significant markup. You end up paying far more for functionality that’s often outsourced anyway, AND you end up locked into a vendor relationship that may not be the best fit for your submission needs.
At Kivo, we believe that publishing is a specialized function that deserves flexibility. Most of our customers already work with trusted publishing vendors or internal teams using tools like Docubridge, and our platform is built to support that model seamlessly. We offer easy publishing exports and eCTD re-import, so your files can move smoothly between Kivo and your preferred publisher, without disruption, rework, or vendor lock-in.
9. What happens if we don’t have a proper RIM system in place during clinical trials?
Without a dedicated RIM system, regulatory teams are forced to rely on spreadsheets, email threads, and disconnected tools to manage high-stakes submissions. This approach may work in the early stages, but it quickly breaks down as timelines accelerate, global requirements multiply, and cross-functional teams grow. The result? Missed deadlines, duplicate work, lost version history, and a serious lack of visibility, especially when working across multiple regions, partners, or contract organizations.
These gaps don’t just slow you down. They expose your company to real regulatory risk. A delayed response to a health authority, a misfiled document, or a missed commitment can trigger everything from agency findings to full clinical trial holds, each of which comes at a cost to your operations, finances, and reputation.
While legacy system have discouraged many smaller teams from transitioning quickly from homegrown data managements to a dedicated RIM system, we developed Kivo to make best-in-class RIM software affordable to life science teams of every size. Our system centralizes your regulatory workflows, tracks every milestone and commitment in real time, and gives your team the tools they need to execute quickly, accurately, and confidently. As your trials get more complex, Kivo RIM helps you stay in control, stay compliant, and stay ahead of regulatory expectations.
10. How long does it take to implement Kivo RIM?
Unlike legacy RIM systems that can take 6–12 months to implement (and require dedicated IT teams), Kivo RIM is designed for fast, low-friction implementation. Many customers are up and running within TWO WEEKS, and our intuitive interface, pre-configured structures, and expert onboarding support make it easy for your team to start seeing value quickly.
We also include lifetime validation and industry-leading support in our pricing, so you’re not hit with surprise fees down the line. Whether you're migrating from a patchwork of tools or replacing a legacy platform, Kivo helps you make the transition smoothly, without disrupting your ongoing submissions or timelines.
Reduce Manual Work and Improve Compliance with the Easiest-to-Use RIM
“I can’t even express how refreshing the ease of use is, because the complexity of the traditional Part 11 Compliant systems is nonsensical. Those were designed by and for large organizations. But Kivo makes it so easy.”
-Head of IT, Elevar Therapeutics