KIVO QMS
A Quality Foundation for Growing Teams
Control your documents, manage quality activities & vendors, and keep track of training with Kivo's validated, Part-11 compliant Quality Management.
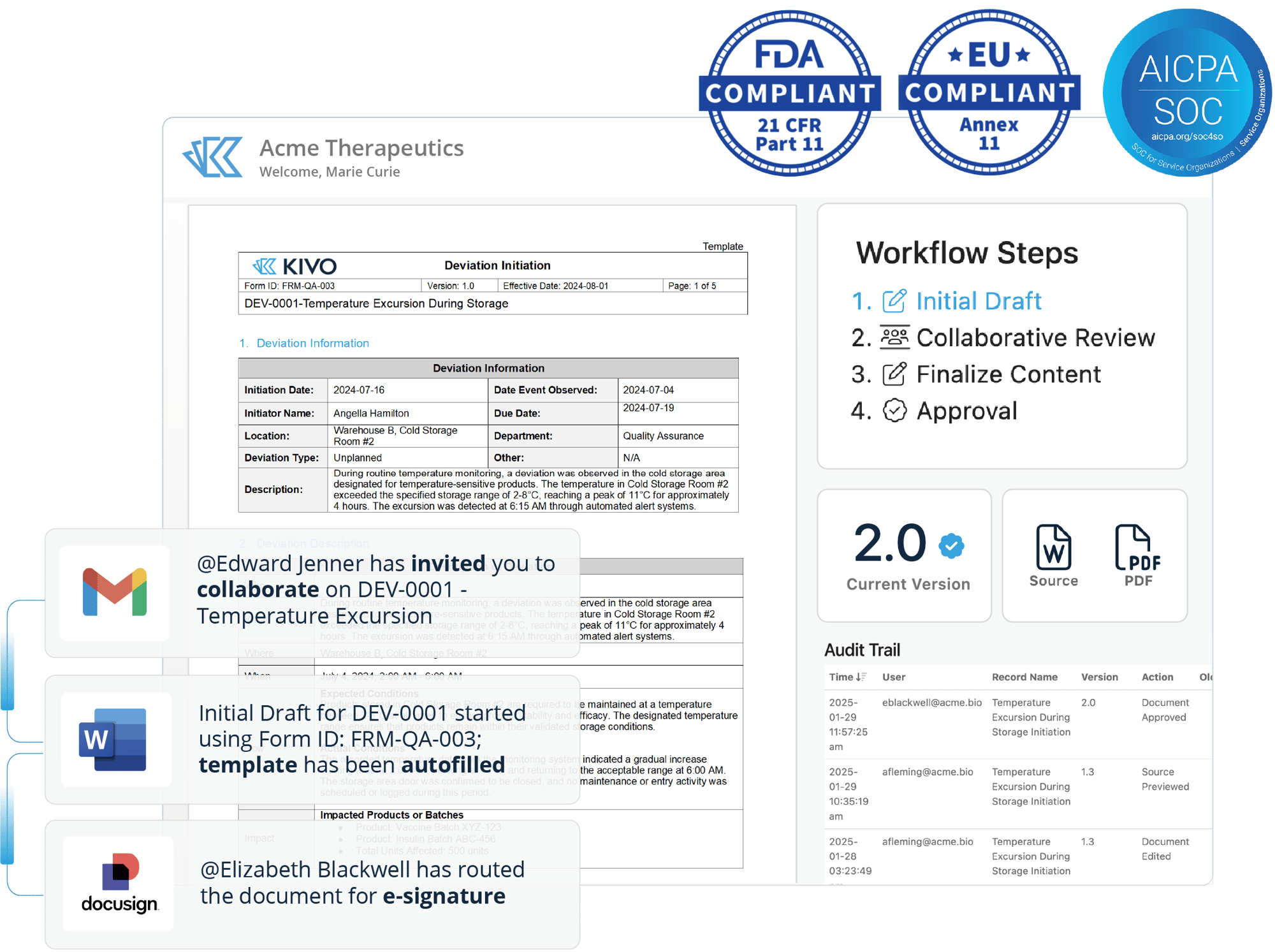
Fully Compliant, Less Complicated
Standardize (& streamline) Quality operations in Kivo’s user-friendly, pre-validated system. Built on our Part-11 compliant Document Management Platform, Kivo’s QMS allows teams to manage their Quality documents and processes without as many clicks.
Confidently Manage Quality Events & Documents
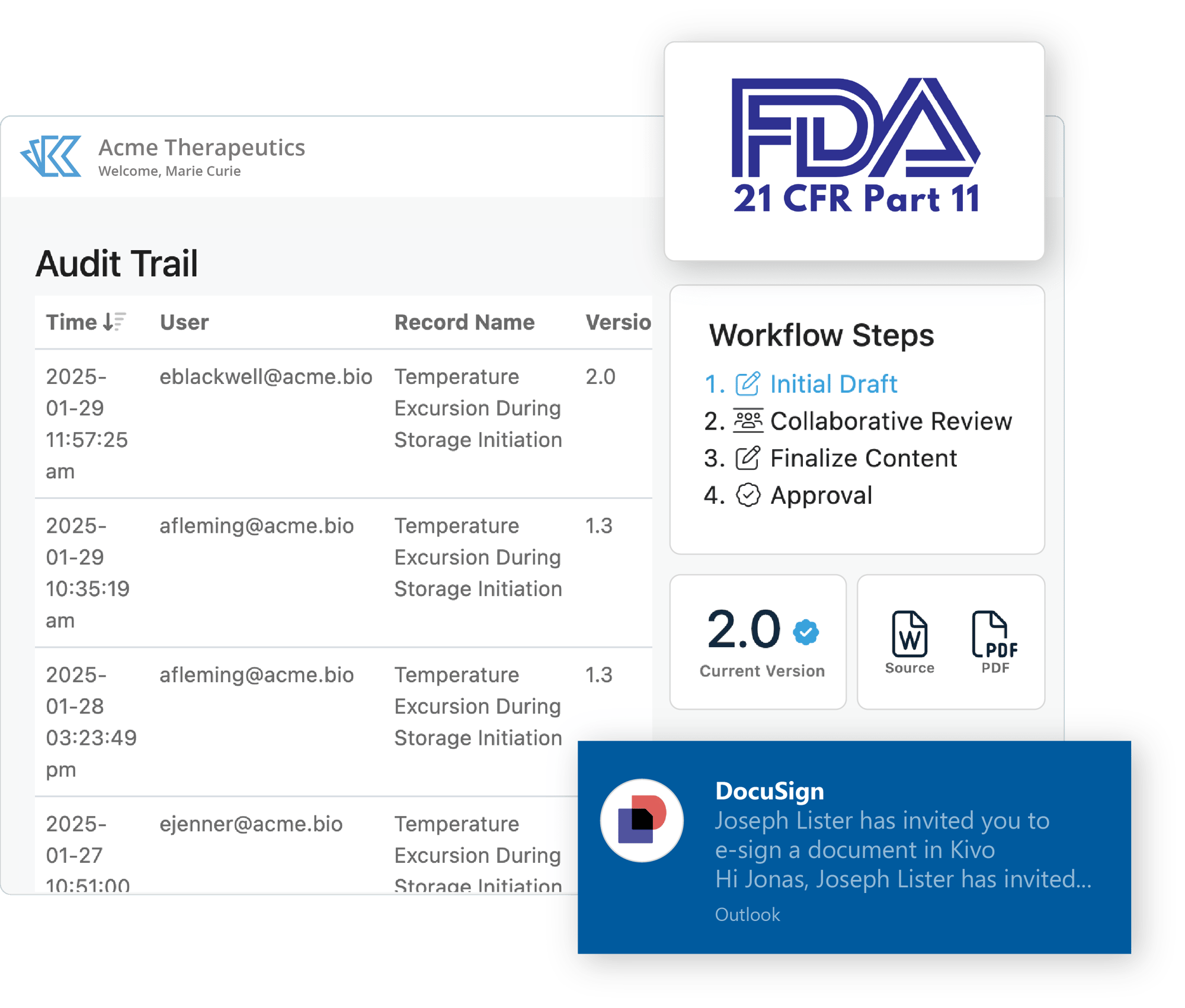
Controlled Documents
Kivo's QMS area leverages our Part-11 compliant Document Management System for authoring, editing, approval of SOPs and other key documents:
- Control user permissions & document access by user role.
- Enforce naming conventions by document type.
- Use templates to maintain consistency and automate document generation.
- Customize your workflows for authoring, editing, review & approval.
- Includes audit-ready 'simple approval' as well as DocuSign for eSignature.
Quality teams also use Kivo's built-in Reporting to track document status, effective & renewal dates, and more.
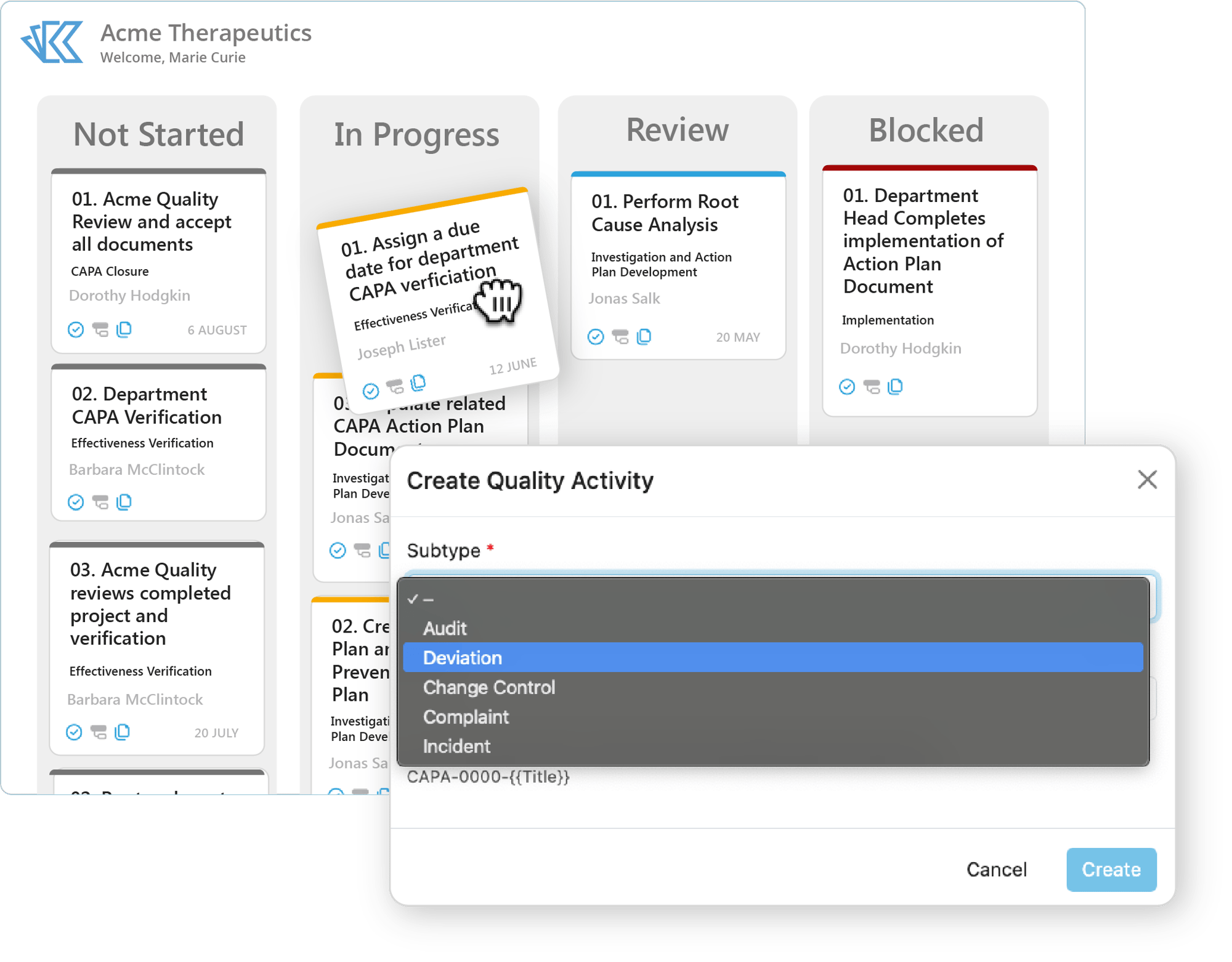
Quality Events
Create, manage, and track all your Quality Events in Kivo. With highly configurable event forms, templates, and workflows - Kivo allows teams to establish compliant quality processes (or to streamline their existing ones).
- Incidents & Complaints
- Deviations & Non-Conformances
- Risk Evaluations
- Change Control Processes
- CAPA Management
Save time with automatically generated documents based on your existing Quality Event templates. Use Kivo's customizable reporting to create dashboards and automatically send reports to your team.
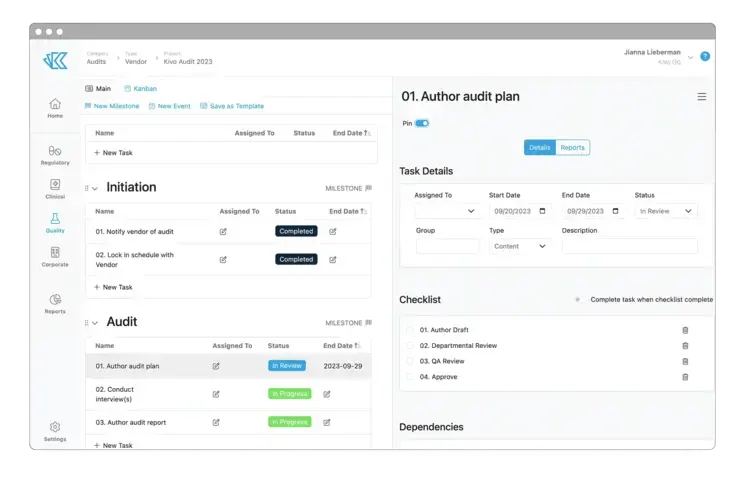
Audits
Simplify and accelerate internal, vendor, and partner audits by leveraging Kivo's QMS.
- With Kivo, your team is always Audit & Diligence-ready. We even include an 'Inspector' user role specifically for remote audits.
- Vendor & Suppliers Audits are easy to manage using our Vendor Management area - with automatically generated forms, documents & even audit project plans.
- Easily link Internal Audits to other Quality Events - such as Risk Evaluations or CAPAs - when needed.
Kivo offers your entire team a Part-11 compliant system system that is always audit-ready, so you can focus on the work at hand.
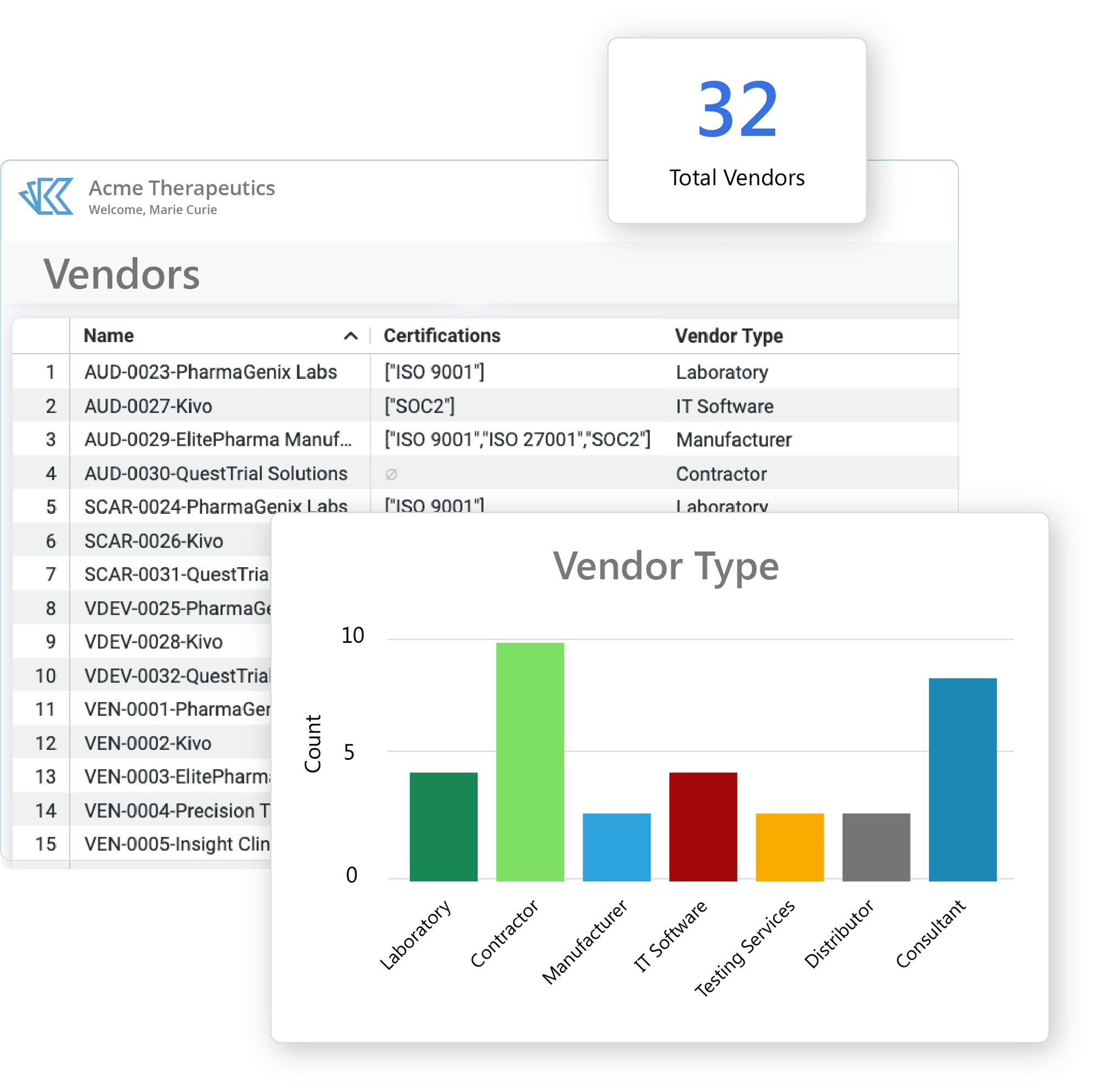
Vendor & Supplier Management
Kivo's QMS is a single system to manage all aspects of your vendor relationships.
- Create & manage all Vendor-related qualification forms and documents.
- Capture key vendor & supplier information and status - including approval status, renewal dates, and more.
- Use Kivo's built-in project management to plan and track regular Vendor activities.
- Leverage Kivo's configurable reports to create vendor dashboards and automatic status reports.
Kivo makes it easy to see and manage all your vendors and suppliers in one place - and stay on top of compliance requirements.
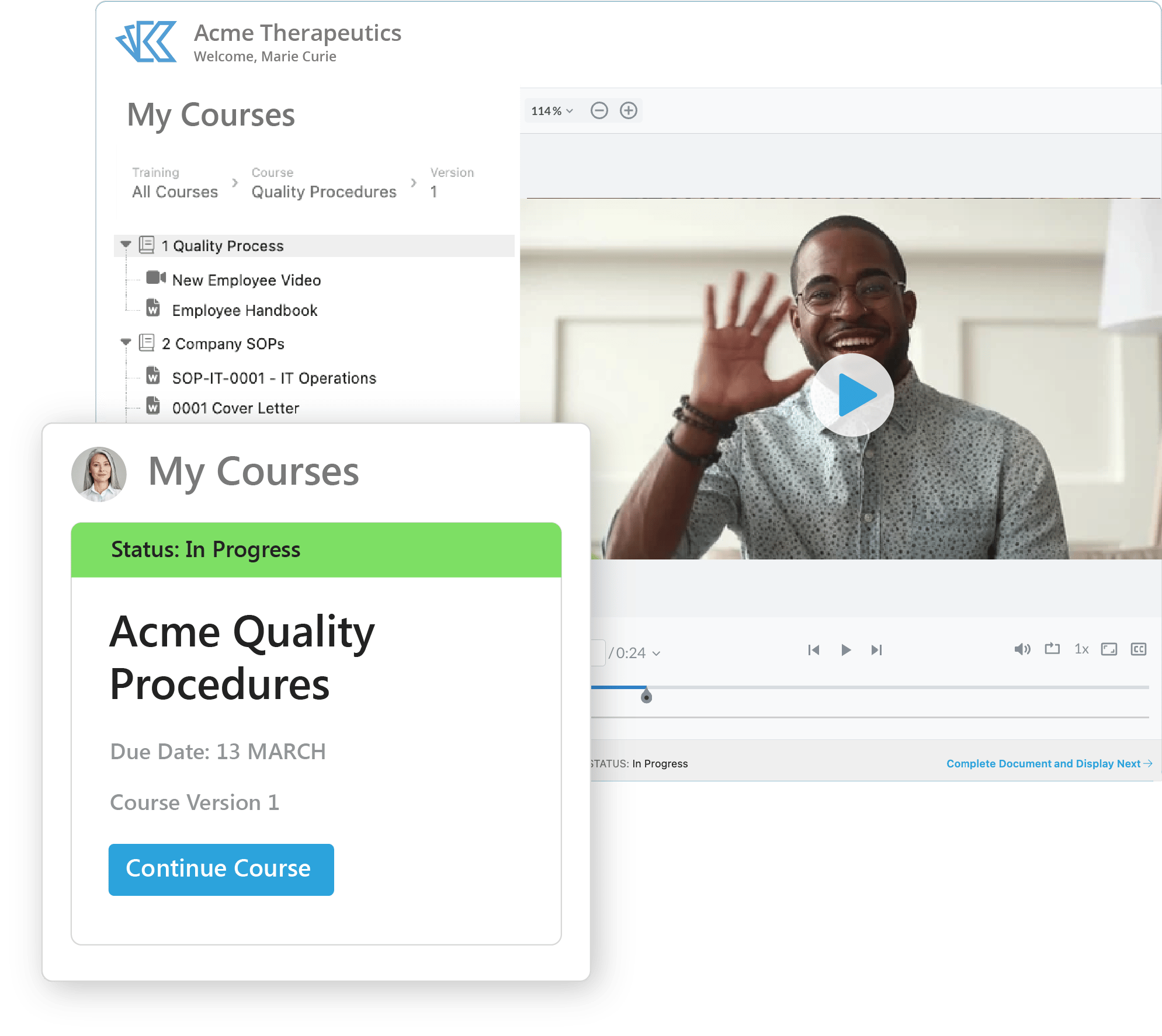
Training Management
It's easy to establish and manage a compliant training program with Kivo's QMS:
- Build Courses from content stored anywhere in Kivo - including SOP docs, videos, and presentations.
- Enroll Users in courses by role / department or individually.
- Use our Training Reports to track and manage training compliance by course, element, and employee.
- Link to external training content and upload training records for storage.
Kivo offers teams a simple and compliant way to manage training on SOPs, corporate policies & more - all as part of our audit-ready QMS.
Best in Class Security & Compliance
Kivo was purpose-built for the compliance requirements of the life sciences industry. We enable GCP/GxP, security & auditability across your org at all times, even conducting quarterly data integrity checks (at no cost to you).
Updates to Kivo's software are pre-validated and delivered with complete documentation. Sponsors around the globe entrust Kivo with their data - and you can too.
Features that scale. Costs that don't.
-
QMS FeaturesApproval Workflows with Part 11 Compliant E-SignaturesSmart Document PlaceholdersPre-Structured & Customizable Project TemplatesSecure Access and ViewingAuto-Associate Projects to Tasks, and Tasks to DocumentsIntelligent Reporting and Automated AlertsLink Trainings Directly to the DMS
-
General Kivo FeaturesAccess to eTMF, eCTD and RIM Included (No Integration Required)Role-Based, Per User Licensing for Full Platform, Limited Access, and 3rd-Party AccessDocument Linking with Aliasing (No Multi-Indexing Required)Seamless Integration with Microsoft Office OnlineCustomizable Metadata Across Features
-
IT & SecurityIntegrated CFR Part 11 Compliant eSignaturesNo IT Required (Fully Cloud Based)Single Sign OnFully Browser Based, Available for PC and Mac
-
SupportLightning Fast Support During Business Hours (Under 5 Minutes!)Unlimited Live TrainingImplement Within WeeksUnlimited Support
Support from Day 1
Implement Within Weeks
Kivo goes through a rigorous validation process to reduce the burden on your organization and reduce implementation timelines. No more implementations that drag on for months - seriously.
Unlimited Training
Receive unlimited training as long as you are a customer. We want to make sure your team is comfortable and able to get work done, so we’ll be there to help every step of the way, even when you bring on new team members.
Real Human Support
Support is available from a real human being 5 days a week from 6am to 6pm PST. Our current response time during those hours is under 5 minutes (really!)
Accelerate Quality Management
Get Kivo QMS today
Kivo QMS FAQs
Kivo QMS (Quality Management System) is a software solution designed to help organizations in the life sciences sector manage their quality processes effectively. Common quality processes include authoring and managing SOPs, overseeing CAPAs, Deviations, and Audits, and enabling required training across the organization and third party vendors.


