Imagine you have assembled your regulatory submission after months (or even years!) of effort. It is now time to send it to a publisher to compile into the correct format for submission to the appropriate regulatory agency. That format is the Electronic Common Technical Document (eCTD), the standard format through which sponsors can submit regulatory information to regulatory agencies around the world. (The current version of the eCTD guidelines, eCTD 4.0, is the latest update to this format).
After your submission has been filed, it is crucial to maintain a copy of the eCTD for compliance and future inspections. Should the regulatory agency (or a potential buyer / investor) have follow up questions regarding your submission, you'll want to be able easily find the documents and data in question. In this case, you'll need an eCTD viewer! And ideally, you'll want one that can help you find the precise document within your document management system.
With Kivo GO, you can do just that, with auto-association of documents from the eCTD to the correct doc in the DMS.
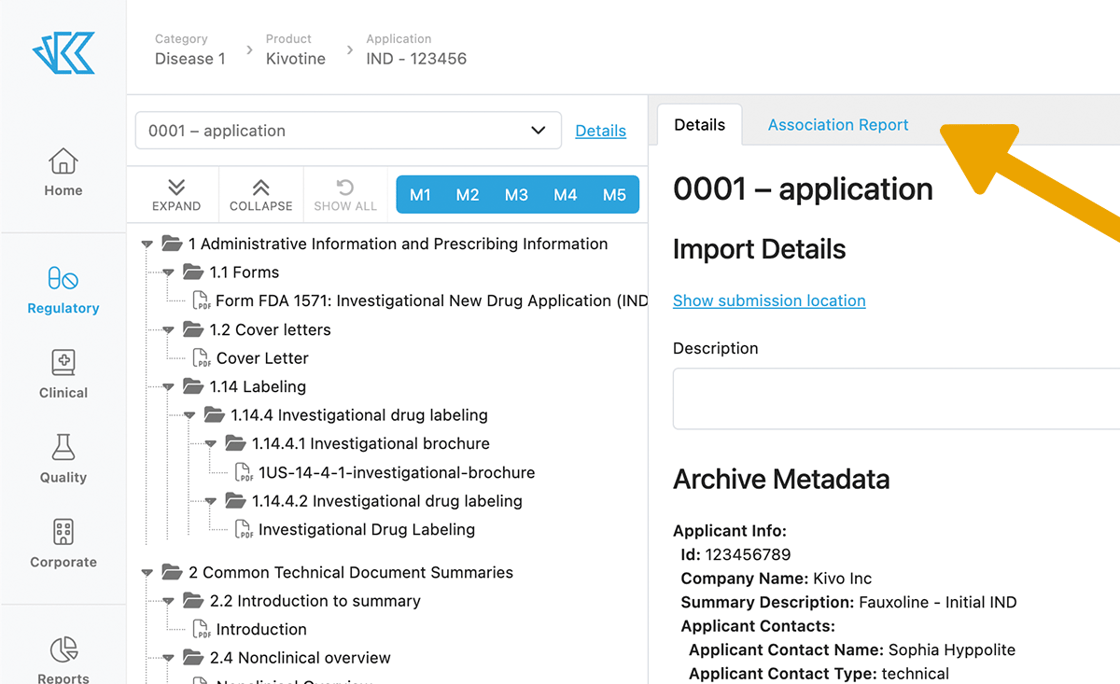
From the eCTD Viewer, you can click on "Association Report" to see all of the documents associated with your submission.
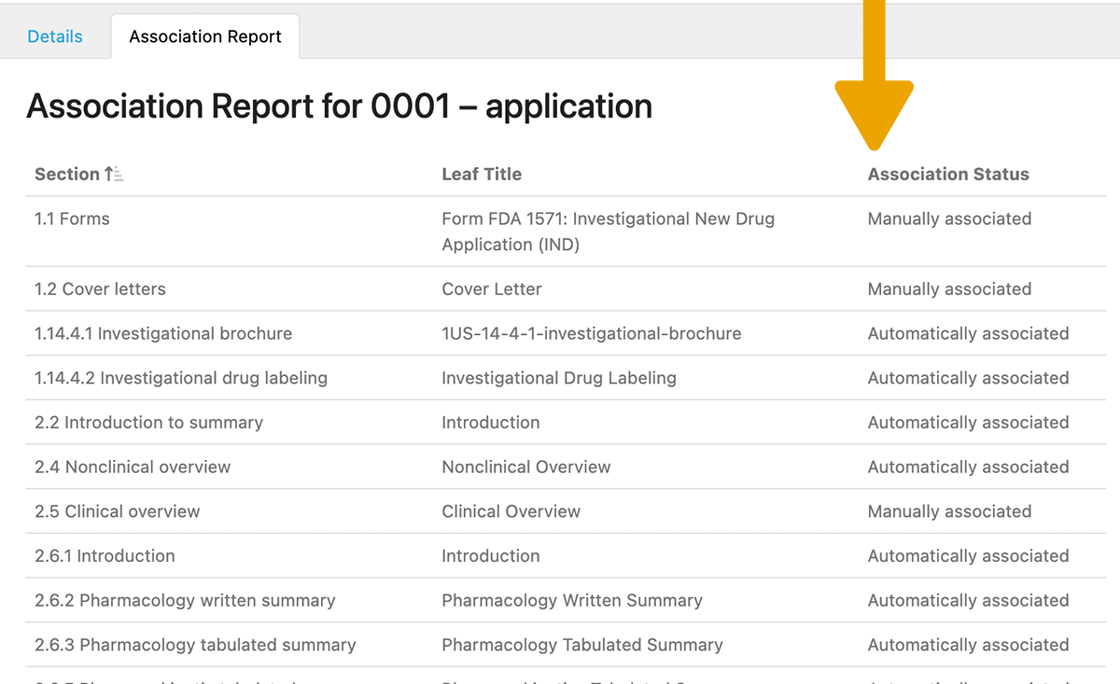
As you can see, many of the documents were automatically associated! You can override these associations manually, but automatic linking saves a ton of time. And, it makes finding the right document down the line effortless.
And that's a wrap! To see more of Kivo's document management capabilities, check out our click-through demo. If your team or colleagues could benefit from a simple, cost-effective DMS solution, reach out to us at any time.


