The Electronic Common Technical Document (eCTD) has long been the backbone of regulatory submissions, streamlining the process of dossier submissions for marketing authorization applications. As the landscape of life sciences continues to evolve, so does the eCTD standard. Enter eCTD V 4.0, the latest iteration set to improve the way regulatory information is presented and accessed. In this article, we delve into what eCTD V 4.0 entails, why this version came into existence, who stands to benefit from it, and the timeline for its implementation, preparing stakeholders for a future of enhanced efficiency and collaboration. In our next article, we'll do a deeper dive on what the changes are and how to incorporate them.
Now let's dive in!
The What: What is eCTD 4.0?
Global Agencies currently accept eCTD v. 3.2.2. The next major version of the eCTD is v. 4.0. It has been in discussion and development for over 10 years. Based on the Regulatory Product Submission (RPS) created by Health Level Seven (HL7), eCTD v. 4.0 introduces elements set up to handle more data driven submissions in the future. Since it is based on the RPS model, other submission types like Veterinary and Medical Devices could be incorporated in the future.
The Why: Why is there a new version of the eCTD?
In a nutshell, the goal of eCTD v.4.0 is to increase speed, flexibility and communication for regulatory submissions for and between agencies and sponsors.
Over the many years of multi-agency and sponsor use of the eCTD, requests and suggestions have been made to increase efficiency on both sides. As the acceptance of XML formats increased, the time has come to accommodate more controlled terms, easier lifecycle and single harmonized communication package.
The latest version, v. 4.0, extends the functionality of XML by making it more flexible in the ways to work with document reuse as well as updates to software for regulatory changes. The ability to change the DTD (document type definition) without updating software is a huge advantage to both agencies and sponsors.
Concepts introduced include Context of Use, ability to order (set priority) and group documents, use of controlled vocabularies (CVs) and document re-use. Each of these concepts allow sponsors to more accurately create and tell a story in their submission for regulators. With eCTD 4.0, more exacting, clear and concise documentation can be provided by the sponsor for analysis at the agency.
The Who: Who does eCTD 4.0 affect?
New submission guidance affects the regulatory teams who submit the package to agencies. Cross functional contributors like medical writers, statisticians, data managers, and CMC will need to have a high-level understanding of the new concepts for tagging their content correctly in the XML. Internal IT teams may be called upon to upgrade current systems- EDMS and regulatory operations publishing solutions to meet the new technical requirements.
The When: When does eCTD 4.0 take affect?
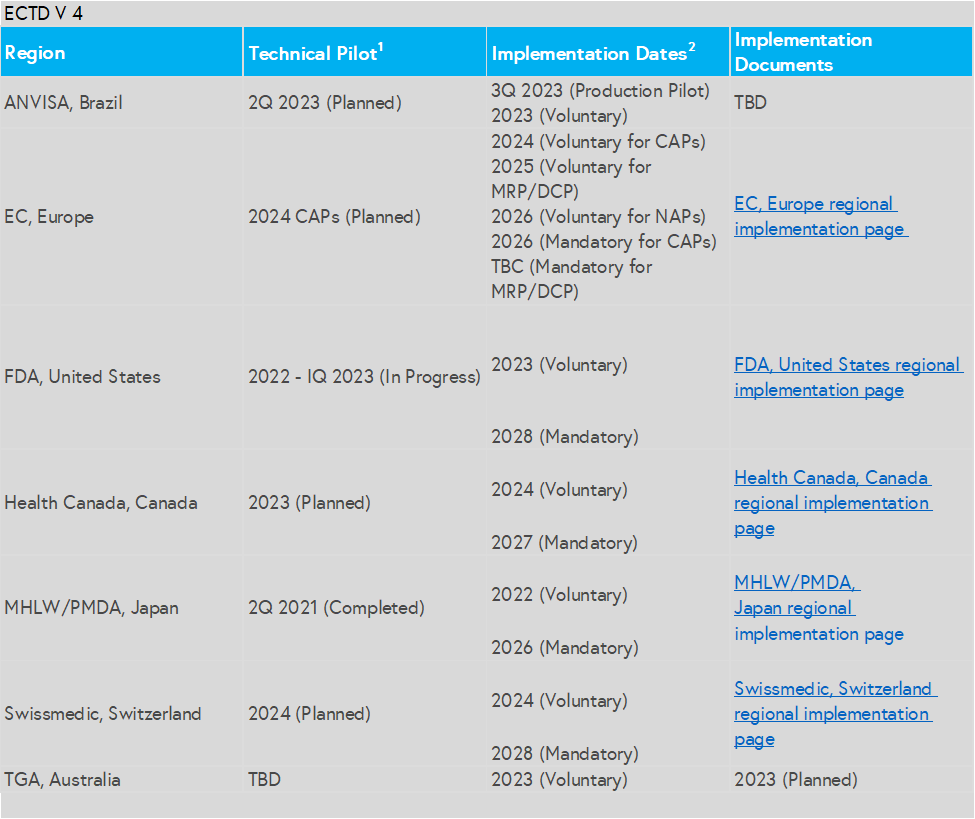
Global agencies are already starting to accept eCTD v 4.0 on a voluntary basis. Japan PMDA began in 2022 and US FDA in early 2023. Although the official cutover dates are a few years in the future, getting operational experience early sets a sponsor up for success in the long term.
Below is a quick view of the Regional Implementation Information: