In pharma and biotech, managing regulatory information effectively is crucial for compliance and success. An Electronic Document Management System (EDMS) provides a powerful solution to streamline and organize regulatory information, ensuring access control, information security, and collaboration.
In this article, we will explore how to leverage an EDMS for regulatory information management (RIM) and highlight the key considerations for its implementation. From compliance requirements to migration considerations, utilizing EDMS for third parties, electronic submissions, and cross-functional collaboration, we will cover various aspects that will help you harness the full potential of an EDMS.
Start with the Goal in Mind
Before diving into the details of using an EDMS for RIM, it is essential to consider your goals and requirements. Analyze your submission workload and the number of team members involved to establish a clear understanding of your needs. One common pitfall is investing in a system that is too complicated, with unnecessary features that may not be utilized for months or even years. To avoid this, focus on the must-have functionalities that align with regulatory compliance.
Compliance Requirements of an EDMS
- Provide access control to documents based on users: An EDMS should offer robust access control mechanisms to ensure that only authorized individuals can access sensitive regulatory documents.
- Provide an electronic record of every action: An audit trail is crucial for compliance purposes. The EDMS should record the person, date, and action taken for each activity within the system.
- Provide centralized storage for data: A centralized storage system allows for efficient management and retrieval of regulatory information, reducing duplication and improving organization-wide access.
- Provide high-level information security: Regulatory information is highly sensitive and requires stringent security measures. The EDMS should offer robust security features to protect data from unauthorized access or breaches.
Essential Functionalities of an EDMS for RIM
To make the most of an EDMS for RIM, it is important to choose a system that offers the following essential functionalities:
- Facilitate information creation, retention, and distribution: An effective EDMS should streamline the creation, retention, and distribution of regulatory information, ensuring seamless collaboration across teams.
- Ensure fast and easy information retrieval: A familiar folder structure based on industry standards such as Electronic Document Management (EDM), International Council for Harmonisation (ICH), or electronic Common Technical Document (eCTD) helps expedite information retrieval. The system should also support powerful search capabilities to quickly locate specific documents.
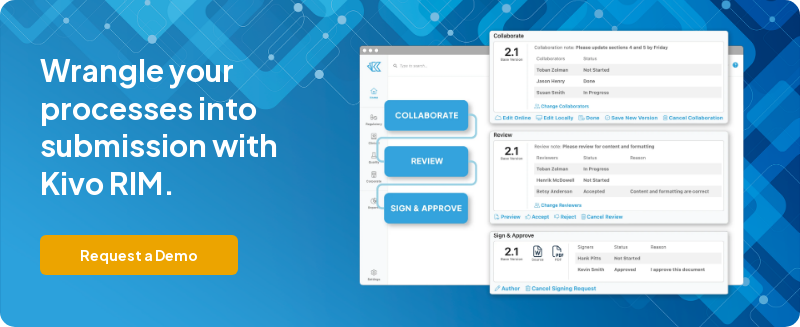
- Support content-driven collaboration: The EDMS should enable multiple users to collaborate on regulatory documents, both online and offline. It should provide simple tools to create new versions, ensuring efficient collaboration throughout the document lifecycle.
- Offer automated workflows designed for RIM: Automated workflows tailored for regulatory information management can streamline processes, improve efficiency, and reduce manual errors.
- Provide pre-built authoring templates following ICH guidance: Pre-built authoring templates aligned with ICH guidance can save time and ensure adherence to regulatory standards during content creation.
- Enable time-saving automation for repeatable folder structures: Automation features that facilitate the creation of repeatable folder structures can save time and enhance consistency in document organization.
- Offer a user-friendly interface: A user interface that is intuitive and easy to understand is essential for user adoption. It should be designed with the end-users in mind to ensure a seamless user experience.
- Support PDF conversion following ICH and FDA guidance: The ability to convert documents to PDF format while adhering to ICH and FDA guidelines is crucial for regulatory submissions.
Ok you've chosen your EDMS 🙌 Now we need to migrate.
So how do you migrate?
When migrating to an EDMS for RIM, it is important to consider the following aspects to ensure a smooth transition:
- Choose a knowledgeable vendor: Select a vendor who specializes in the regulatory and clinical industry. Ensure they have a deep understanding of the regulatory landscape to provide the necessary expertise and support during the implementation process. Having the right partner will make the process significantly easier!
- Build you content migration plan: Develop a comprehensive plan that includes migrating data, roles, and responsibilities from existing systems to the new EDMS.
- Define your validation and implementation timelines: Align the validation and implementation timelines with your submission schedule to avoid any disruptions or delays in regulatory processes.
- Provide a QC/QA process: Implement a quality control and quality assurance process to ensure data integrity and accuracy during the migration process.
- Map metadata: Properly map metadata from the existing system to the EDMS to ensure seamless information retrieval and consistency.
- Train your team: Provide comprehensive training to users to familiarize them with the new EDMS and its features, ensuring smooth adoption and efficient utilization. Depending on your vendor, they may offer a set number of training sessions or hours. Some vendors, like Kivo, offer unlimited live training.
Once you're migrated, it's time to start using your new system!
In today's world of decentralized clinical trials and electronic submissions, this means learning to manage resources from afar. Let's start with submissions.
Using EDMS for Electronic Submissions
RIM plays a crucial role in facilitating regulatory electronic submissions. Emerging pharmaceutical companies often outsource regulatory writing and publishing work. If you are not hosting your regulatory submission sequences, it is essential to plan for a compliant transfer from your regulatory publishing vendor after agency acceptance. As the sponsor, you are responsible for knowing what you submitted to the reviewers and being able to respond to questions or requests for information. Importing and viewing the eCTD sequences in your RIM system allows your team to stay informed about the submission details and respond promptly to reviewer inquiries.
Re-using content within submissions is common practice. Subject matter experts determine which version of content to use based on region and submission type. An EDMS, such as Kivo EDMS, provides visibility into the document's location and the version used, which becomes extremely helpful as submissions grow and content re-use becomes more complex.
Utilizing EDMS for 3rd Parties
Collaborating with third-party contractors, such as Contract Manufacturing Organizations (CMOs), Contract Research Organizations (CROs), Medical Writers (MWs), and regulatory operations partners, can bring numerous benefits in terms of cost reduction and increased productivity. Leveraging a powerful EDMS can streamline the handoffs, improve oversight, and facilitate project management. Here are some key considerations for utilizing EDMS for third parties:
- Define your vendor handoff approach: Clearly define roles and responsibilities, along with timelines for activities, when working with third-party vendors.
- Document access and review in Contracts, SOWs, and Project Plans: Specify how and where documents will be accessed, reviewed, and approved in your contracts, statements of work, and project plans.
- Real-time review of document metrics: Monitor document authoring, review, approval, and publishing metrics in real time to ensure efficient collaboration and accountability.
- Drive continuous process improvement: Act upon the data gathered through the EDMS, hold individuals accountable, and drive continuous process improvement across your regulatory processes.
Using EDMS cross-functionally with Clinical Operations
In the world of regulatory information management, critical documents often have links to Trial Master Files (TMFs) or Clinical Project Milestones. By leveraging an EDMS, you can establish triggers, alerts, and events within project plans to ensure seamless collaboration and adherence to project deadlines. For example, the "Letter of Authorization" in Regulatory IND or CTA is the official authorization to start a clinical trial. Your clinical colleagues will need this document filed within the TMF in the “Trial Approval” section and Artifact 3.01.02 Regulatory Authority Decision. You’ll want to file this document and alert your clinical team. Furthermore, protocols, statistical analysis plans, and other key documents generated or filed in the TMF by the clinical development and biostatistics teams play a vital role in regulatory submissions. An EDMS that provides alerts when the approved versions are ready can save significant time and mitigate potential challenges during critical submission periods.
Conclusion
As organizations venture into their first agency submissions, the need for a robust RIM system becomes paramount. While basic storage and collaboration tools like SharePoint may suffice for initial stages, the requirements evolve as compliance, control, and efficiency take center stage. Implementing an EDMS for regulatory information management empowers teams to meet regulatory goals, ensure compliance, and streamline processes. By considering compliance requirements, leveraging essential functionalities, addressing migration considerations, utilizing EDMS for third-party collaborations, optimizing electronic submissions, and achieving cross-functional document excellence, organizations can harness the power of an EDMS and elevate their regulatory information management practices. Stay ahead of the curve and be the winner in your organization by embracing an EDMS that aligns with your regulatory goals and helps you achieve success.