One of the more challenging aspects of clinical trial management is visibility. The sheer number of outsourced partners - sites, CROs, writers, consultants, vendors, etc - who often have their own systems and workflows for communication and collaboration makes it difficult to know if your projects are on track.
By managing your content within the same system as your reporting, you can enable real-time visibility into your operations. At Kivo, that's precisely what we've designed. Let's take a look at how it works!
Reports are accessible from your lefthand side menu. Reports can be built for your specific needs, but we offer a suite of pre-built reports for a variety of Clinical, Regulatory, and Quality needs.

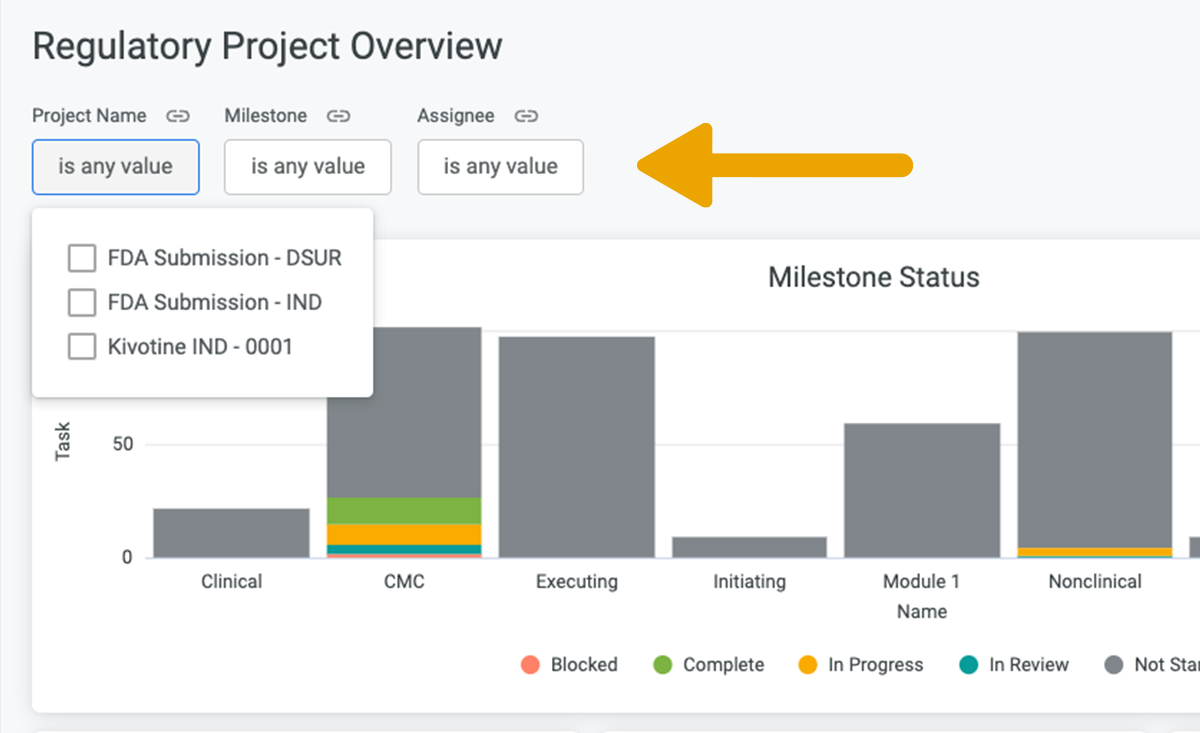
For the purposes of this example, let's look at the Regulatory Project Overview . This is a central report that summaries the status of all your Regulatory Projects by Milestone and Assignee, making it easy to see if your submissions are on track.

Filter the report as needed to get instant visibility into each Project.

To make life even easier, you can Schedule Delivery of your most used Reports directly to your inbox! Let's check out the available options.

You can choose how often you'd like to receive an email, who should receive it, and in what format. We currently support PDF, CSV, and PNG formats. You can also filter the delivery to include a single project or specific milestones. You could also deliver a weekly report to an assignee, for example, so they get a summary of everything they need to work on.

You can Test now to see how the report will look on the receiving end, and hit Save if everything looks good. Then getting updates on your Projects is as simple as checking your email.
Less work?? We love that for you 😎
If you'd like to see more of our reporting capabilities and how we can reduce the time you spend tracking your work, reach out to us at any time! And to get more info about Kivo in your inbox each month, subscribe below.