Kivo is excited to announce the release of Kivo eTMF – an eTMF solution for emerging life science companies built on the Kivo platform. Kivo eTMF provides companies with a simple path to compliance for managing Trial Master Files with investigator sites and through virtual inspections.
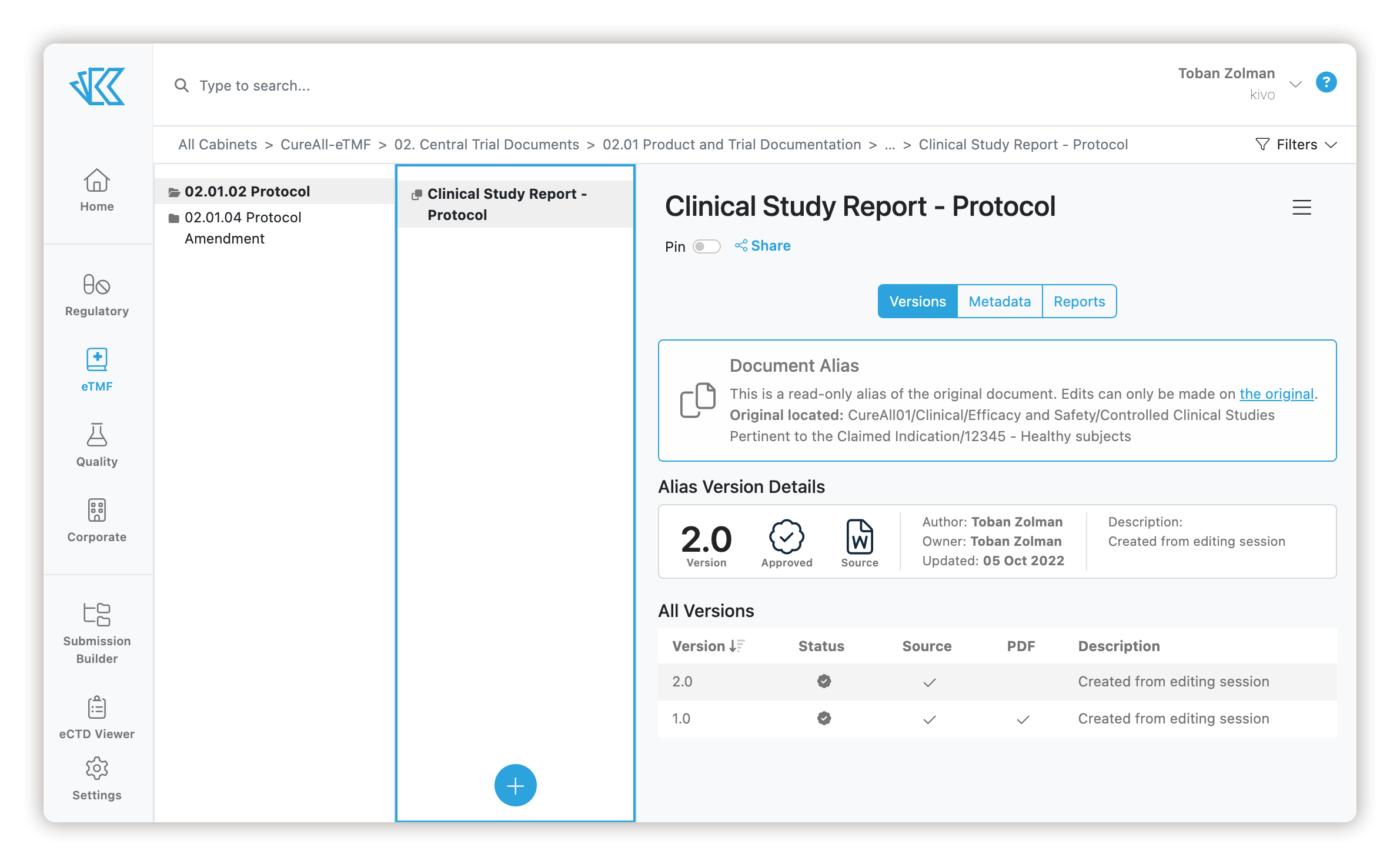
The Intersection of Clinical and Regulatory
Kivo's powerful regulatory platform and EDMS create the perfect foundation for managing clinical documentation. All of Kivo's existing tools work seamlessly in the eTMF product.
Start writing content like the protocol from one of Kivo's document templates. Then, simultaneously edit the document with colleagues and use Kivo's powerful workflow to Review and Approve the document. The approved document can automatically appear in your eTMF. When you submit that protocol to regulators, Kivo will track it across dossiers and provide you with a transparent view of everywhere the document is used, whether you are viewing the document in the DMS, in the eTMF, or in an eCTD. Kivo provides 360-degree visibility into all of your content.
Clinical Document Management that just makes sense
Documents are stored using the eTMF reference model, making navigating and managing content intuitive. With Kivo, you can manage a single authoring and review process and link relevant documents between Regulatory and Clinical in the eTMF and have those links automatically update if a new version is approved.
Your eTMF can be built with placeholders for expected documents, as well as the ability to assign those documents to users and set dates on when they are expected.
Virtual Inspections Made Easy
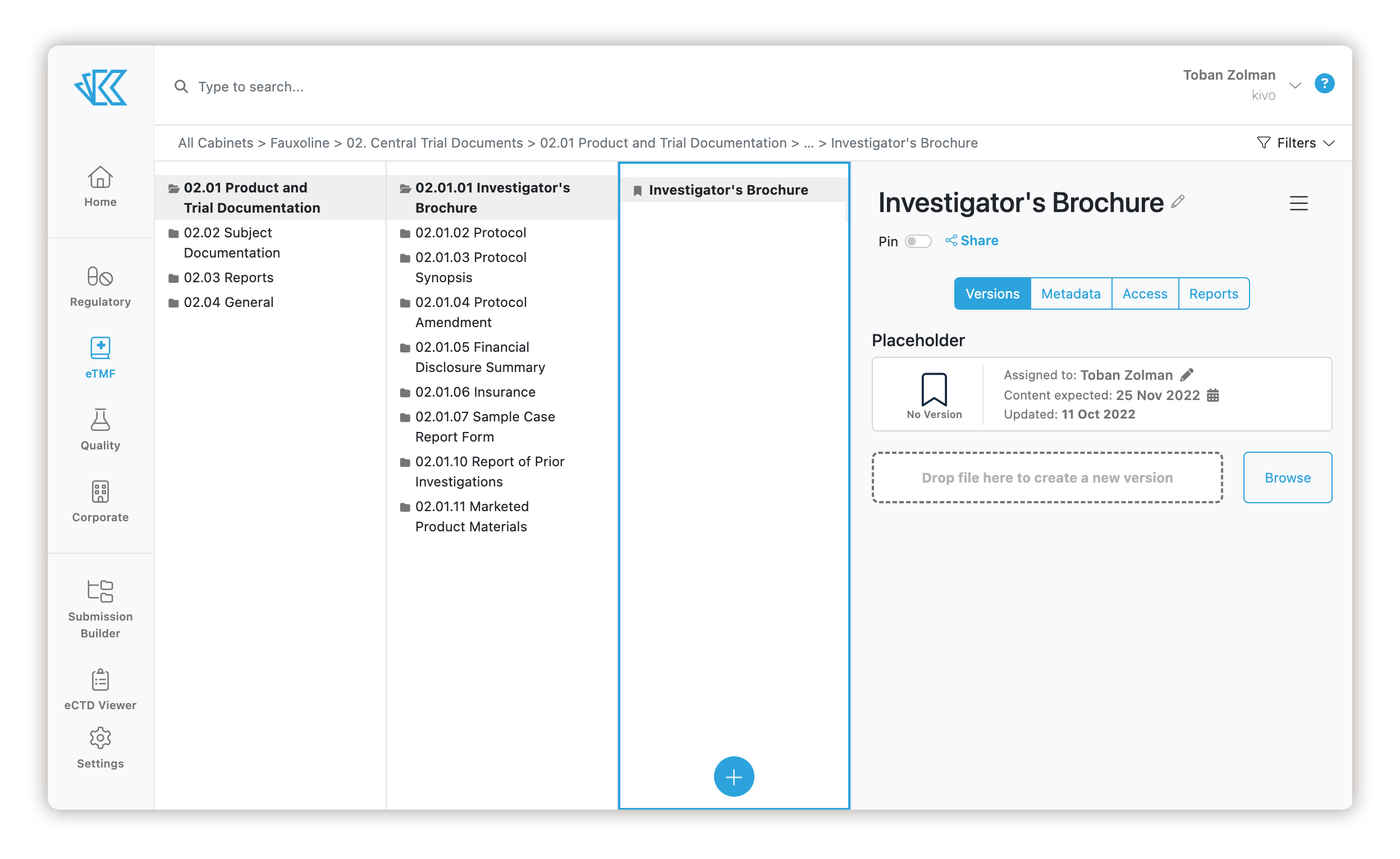
Kivo was designed from the ground up to support virtual inspections. Inspectors use a secure account that enables them to view relevant approved documents and understand when missing content is expected. Allow inspectors to the view content without downloading, printing or copying content out of the system.
Easily Manage Studies, Sites, and More
Kivo eTMF makes it easy to manage studies, control sites, and investigators. Our flexible permissions model enables you to quickly add users, expose the appropriate content, and set permissions. Enable partners and Sites to upload their deliverables but limit their permissions on other content.
Part-11 Compliant, EMA CTIS-ready
Kivo is cloud-based, with nothing to install for Sponsors, Sites, or Investigators. Provided pre-validated, Kivo can be implemented and up and running within days without extensive professional services or configuration.
Request a demo to learn how Kivo can transform your organizational processes and accelerate your time to approval.



