Emerging life science organizations often deal with authoring processes that are lengthy and complicated. Documents are difficult to format and maintain, and users often spend the bulk of their time figuring out what content is expected by a health authority and fighting formatting issues.
Enter Kivo.
Out of the box, Kivo delivers instant access to ICH granular document templates. By centralizing all aspects of the authoring and review process in a single cloud-based tool, Kivo is a sophisticated solution to manage regulated content.
Document authorship can be costly and complex.
Understanding how to write ICH documents can be a tall order even for seasoned biotech organizations. With so many different types of submissions, each with its own set of regulations, it's easy for your resources to be diverted into time-consuming tasks rather than what matters most: the ongoing authorship process.
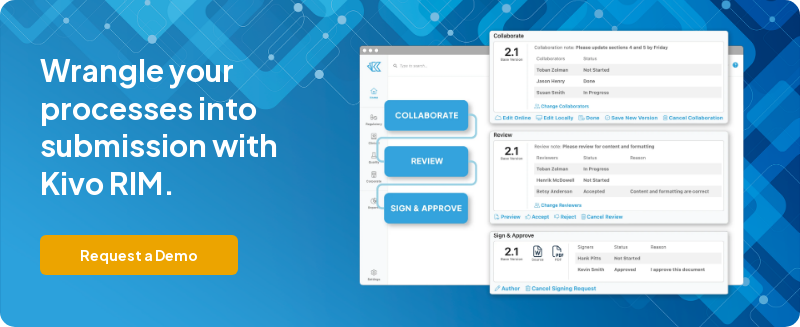
Kivo’s regulatory eCTD packages are designed with distributed teams in mind. Control access to documents, set permissions, and get the right content in the hands of the right person. Kivo’s templates do not require local installation, individual licenses, or macros to use. Authors can edit in Word desktop or Office 365 online making it an excellent choice to collaborate with partners and contractors.
Kivo’s templates simplify the process, ensuring your documentation aligns with FDA, EMA, or Health Canada guidelines.
Kivo templates propel collaboration, compliance and consistency.
Equipped with over 300 ICH and regional document templates, Kivo puts content control back into your hands. Each template was built from the ground up to include features and instructions that simplify document formatting and ensure consistency:
-
No local installation is required.
-
ICH granularity in Modules 2-5.
-
Stand-alone documents are editable from a variety of environments.
-
Region and submission specific instructional text is visible only to content developers.
-
Pre-built TOCs, listings, numbering conventions, tables, sections, file naming, and headings.
-
Support solutions to repair issues through our Document Repair Service.

Acting as a single source of truth for compliance and document management, Kivo streamlines the workflow from authorship to submission.
Kivo is the authoring tool for submissions that get accepted. Schedule your demo with a member of our team today.